An Informational Outline of the Controlled Substances Act
for prescribing, administering, and dispensing controlled substances under the Controlled Substances Act (CSA), Title 21, United States Code (21 U.S.C.) 801-971 and DEA regulations, Title 21, Code of Federal Regulations (21 CFR), Parts 1300 to End. This Pharmacist’s Manual is not a legal document. It is a guidance document that
Substance, Controlled, Dispensing, Controlled substances, Dispensing controlled substances
Download An Informational Outline of the Controlled Substances Act
Information
Domain:
Source:
Link to this page:
Please notify us if you found a problem with this document:
Documents from same domain
Controlled Substances Act - Justice
www.deadiversion.usdoj.govDrug Enforcement Administration . Pharmacist’s Manual
BENZODIAZEPINES (Street Names: Benzos, …
www.deadiversion.usdoj.govDrug Enforcement Administration Office of Diversion Control Drug & Chemical Evaluation Section BENZODIAZEPINES (Street …
Administration, Enforcement, Drug, Drug enforcement administration
Controlled Substances Act - deadiversion.usdoj.gov
www.deadiversion.usdoj.govUnited States Department of Justice Drug Enforcement Administration Office of Diversion Control . P. harmacist’s . M. anual. An Informational Outline of the
Department of Justice - deadiversion.usdoj.gov
www.deadiversion.usdoj.govVol. 79 Tuesday, No. 174 September 9, 2014 Part II Department of Justice Drug Enforcement Administration 21 CFR Parts 1300, 1301, 1304, et al.
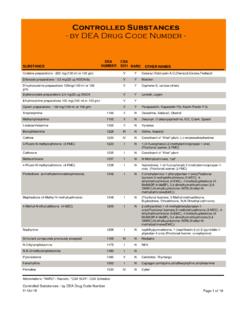
Controlled Substances by DEA Drug Code Number
www.deadiversion.usdoj.govControlled Substances - by DEA Drug Code Number - DEA CSA SUBSTANCE NUMBER SCH NARC OTHER NAMES Codeine preparations - 200 mg/(100 ml or 100 gm) V Y Cosanyl,Robitussin A-C,Cheracol,Cerose,Pediacof
Drug, Substance, Controlled, Controlled substances by dea drug
Scheduling Actions Controlled Substances …
www.deadiversion.usdoj.govScheduling Actions - Alphabetical Order - FINAL ORDER SUBSTANCE PROPOSAL FEDERAL *Scheduled under 21 USC 811(h) PUBLICATION PUBLICATION REGISTER EFFECTIVE CSA
Scheduling, Action, Substance, Controlled, Scheduling actions controlled substances
APPLICATION FOR REGISTRATION APPROVED OMB …
www.deadiversion.usdoj.govForm-225 APPLICATION FOR REGISTRATION Under the Controlled Substances Act APPROVED OMB NO 1117-0012 FORM DEA-225 (04-12) FORM EXPIRES: 7/31/2018 INSTRUCTIONS Save time - apply on-line at www.deadiversion.usdoj.gov DEA OFFICIAL …
Form, Applications, Under, Substance, Controlled, Usdoj, Under the controlled substances act, Deadiversion
An Informational Outline of the Controlled …
www.deadiversion.usdoj.govThis manual has been prepared by the Drug Enforcement Administration, Office of Diversion Control, to assist practitioners (physicians, dentists, veterinarians, and other
Outline, Informational, Controlled, Informational outline of the controlled
U. S. DEPARTMENT OF JUSTICE – DRUG …
www.deadiversion.usdoj.govForm DEA-41 See instructions on reverse (page 2) of form. OMB APPROVAL NO. 1117-0007 Expiration Date 10/31/2020 U. S. DEPARTMENT OF JUSTICE – DRUG ENFORCEMENT ADMINISTRATION
Mid-Level Practitioners Authorization by State
www.deadiversion.usdoj.govMid-Level Practitioners Authorization by State Pursuant to Title 21, Code of Federal Regulations, Section 1300.01(b28), the term mid-level practitioner means an individual
States, Levels, Practitioner, Authorization, Mid level practitioners authorization by state
Related documents
CHAPTER 19-03.1 UNIFORM CONTROLLED SUBSTANCES …
ndlegis.govUNIFORM CONTROLLED SUBSTANCES ACT 19-03.1-01. Definitions. As used in this chapter and in chapters 19-03.2 and 19-03.4, unless the context otherwise requires: 1. "Administer" means to apply a controlled substance, whether by injection, inhalation, ingestion, or any other means, directly to the body of a patient or research subject by: a.
Prescriptions for Controlled Substances
www.deadiversion.usdoj.govPartial Fills of Schedule II Controlled Substances. amended Title 21, United States Code, Section 829 (21 U.S.C. § 829), by adding subsection (f), which states that a "prescription for a controlled substance in schedule II may be partially filled" at the request of the patient (e.g., ultimate user) or… Partial Filling of Prescriptions for
Prescription, Substance, Controlled, Controlled substances, Prescriptions for controlled substances
Part 80: Rules and Regulations on Controlled Substances in …
www.health.ny.govPart 80: Rules and Regulations on Controlled Substances in NYS Effective Date: 02/09/94 Title: Section 80.3 - Exceptions, reclassification and exemptions of scheduled controlled substances 80.3 Exceptions, reclassification and exemptions of scheduled controlled substances. (a) Exemptions. (1) Those nonnarcotic substances listed in section 1308.22 of title 21 of the Code …
Pennsylvania Code
www.health.pa.govJan 04, 1984 · CHAPTER 25 CONTROLLED SUBSTANCES, DRUGS, DEVICES, AND COSMETICS GENERAL PROVISIONS § 25.1. Definitions. The following words and terms when used in this chapter, have the following meanings, unless the context clearly indicates otherwise: Act—The Controlled Substance, Drug, Device and Cosmetic Act (35 P. S. § § 780-101— 780 …
Controlled Substances (Poisons)Regulations 2011
www.legislation.sa.gov.auControlled Substances (Poisons) Regulations 2011 . under the . Controlled Substances Act 1984. Contents . Part 1—Preliminary. 1 Short title. 3 Interpretation. ... Part 4—Prescriptions and dispensing. 33 How prescriptions are to be given. 34 Written prescriptions. 34A Giving prescriptions for monitored drugs—special provisions.
Part 2640: Prescribing, Administering and Dispensing
www.msbml.ms.govA. “Administer”, “Controlled Substances”, and “Ultimate User” shall have the same meaning as set forth in Mississippi Code, Section 41-29-105, unless the context otherwise requires. ... dispensing, or prescribing controlled substances in any schedule, said licensee shall be prohibited from registering with the U.S. Drug ...
Part, Prescribing, Substance, Controlled, Administering, Dispensing, Controlled substances, 2406, Part 2640, Administering and dispensing
Chapter 45H Controlled Dangerous Substances
www.njconsumeraffairs.govc) Dispensers of controlled dangerous substances or practitioners registered to conduct research with controlled dangerous substances shall pay an annual fee of $40.00 at the time of application for registration or for renewal of registration. d) Incorporated humane societies or licensed animal control facilities registered to purchase
Chapter, Dangerous, Substance, Controlled, Chapter 45h controlled dangerous substances