GUIDELINES FOR BIOAVAILABILITY & …
GUIDELINES FOR BIOAVAILABILITY & BIOEQUIVALENCE STUDIES Central Drugs Standard Control Organization, Directorate General of Health Services, Ministry of Health & Family Welfare,
Download GUIDELINES FOR BIOAVAILABILITY & …
Information
Domain:
Source:
Link to this page:
Please notify us if you found a problem with this document:
Documents from same domain
SCHEDULE M - Central Drugs Standard Control …
www.cdsco.nic.in[SCHEDULE M] [See Rules 71, 74, 76 and 78] GOOD MANUFACTURING PRACTICES AND REQUIREMENTS OF PREMISES, PLANT AND EQUIPMENT FOR PHARMACEUTICAL PRODUCTS. Note: - To achieve the objectives listed below, each licensee shall evolve appropriate
Good, Practices, Pharmaceutical, Schedule m, Schedule, For pharmaceutical
GUIDANCE FOR INDUSTRY ON PREPARATION OF …
www.cdsco.nic.inguidelines on common technical document (ctd) 28.10.2010 page 1 of 110 guidance for industry on preparation of common technical document for import / manufacture and marketing approval
Document, Guidance, Technical, Preparation, Industry, Common, Guidance for industry on preparation of, Common technical document, Guidance for industry on preparation of common technical document
Ministry of Health and Family Welfare, …
www.cdsco.nic.in1 Ministry of Health and Family Welfare, Government of India has approved 25 panels of experts of various therapeutic areas. For evaluation of various categories of applications of clinical trials, new
Health, Family, India, Government, Welfare, Ministry, Government of india, Ministry of health and family welfare
CDSCO - Guidance for Industry
www.cdsco.nic.inGuidance for Industry Central Drugs Standard Control Organization Page 3 OBJECTIVE This Guidance has been developed in conformity with Drugs and Cosmetics
No.DCG (I/Misc./2017 (68) Directorate General of …
www.cdsco.nic.inPage 1 of 1 No.DCG (I/Misc./2017 (68) Central Drugs Standard Control Organisation Directorate General of Health Services Office of Drugs Controller General India
Central Drugs Standard Control Organization
www.cdsco.nic.inCentral Drugs Standard Control Organization Directorate General of Health Services, Ministry of Health and Family Welfare, Government of India
Guidelines on Similar Biologic: Regulatory …
www.cdsco.nic.inGuidelines on Similar Biologic: Regulatory Requirements for Marketing Authorization in India
Guidelines, Requirements, Regulatory, Marketing, Authorization, Biologics, Regulatory requirements for marketing authorization
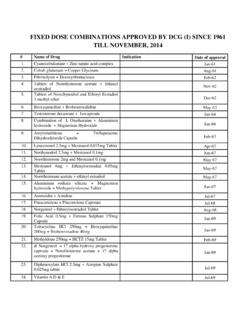
FIXED DOSE COMBINATIONS APPROVED BY DCG …
www.cdsco.nic.inFIXED DOSE COMBINATIONS APPROVED BY DCG (I) SINCE 1961 TILL NOVEMBER, 2014 # Name of Drug Indication Date of approval …
Dose, Till, Combination, Approved, Fixed, Fixed dose combinations approved by dcg
DRAFT GUIDANCE ON APPROVAL OF CLINICAL …
www.cdsco.nic.inguidelines on approval of clinical trial & new drugs effective date: page 1 of 71 draft guidance on approval of clinical trials & new drugs draft guidance
Drug, Guidance, Clinical, Trail, Approval, Guidance on approval of clinical, New drugs, Guidance on approval of clinical trials amp new drugs
CENTRAL DRUGS STANDARD CONTROL …
www.cdsco.nic.incentral drugs standard control organisation directorate general of health services, ministry of health and family welfare government of india
General, Standards, Control, Drug, Central, Organisation, Directorate, Central drugs standard control, Central drugs standard control organisation directorate general
Related documents
Magnesium Absorption and Assimilation - Atrial …
afibbers.orgMagnesium Absorption and Assimilation By Jackie Burgess RDH (ret) SUMMARY Optimizing intracellular magnesium is first step in maintaining normal sinus rhythm.
Medicinal cannabis: Rational guidelines for dosing
cannabisplus.netMedicinal cannabis: Rational guidelines for dosing Gregory T Carter 1*, Patrick Weydt 2, Muraco Kyashna-Tocha 3 & Donald I Abrams 4 Addresses
Guidelines, Medicinal, Cannabis, Rational, Dosing, Medicinal cannabis, Rational guidelines for dosing
Technologies to Improve the Solubility, Dissolution …
medcraveonline.comJournal of Analytical & Pharmaceutical Research Technologies to Improve the Solubility, Dissolution and Bioavailability of Poorly Soluble Drugs
Food system strategies for preventing micronutrient ...
www.fao.orgThe designations employed and the presentation of material in this information product do not imply the expression of any opinion whatsoever on the part of the Food ...
Chapter 5
www.who.intintakes lower than the minimal normative zinc intake, and of 15 surveys conducted in populations with low zinc bioavailability, none reported mean intakes greater than the minimal normative zinc intake.
Bioavailability / Bioequivalence - bebac.at
bebac.atStudy Designs Single Dose / Multiple Dose Standard 2×2 Cross-over Parallel Groups for more than 2 Formulations Bioavailability / Bioequivalence 2 Study Designs (Single Dose / …