SCHEDULE M - Central Drugs Standard Control …
[SCHEDULE M] [See Rules 71, 74, 76 and 78] GOOD MANUFACTURING PRACTICES AND REQUIREMENTS OF PREMISES, PLANT AND EQUIPMENT FOR PHARMACEUTICAL PRODUCTS. Note: - To achieve the objectives listed below, each licensee shall evolve appropriate
Download SCHEDULE M - Central Drugs Standard Control …
Information
Domain:
Source:
Link to this page:
Please notify us if you found a problem with this document:
Documents from same domain
GUIDANCE FOR INDUSTRY ON PREPARATION OF …
www.cdsco.nic.inguidelines on common technical document (ctd) 28.10.2010 page 1 of 110 guidance for industry on preparation of common technical document for import / manufacture and marketing approval
Document, Guidance, Technical, Preparation, Industry, Common, Guidance for industry on preparation of, Common technical document, Guidance for industry on preparation of common technical document
Ministry of Health and Family Welfare, …
www.cdsco.nic.in1 Ministry of Health and Family Welfare, Government of India has approved 25 panels of experts of various therapeutic areas. For evaluation of various categories of applications of clinical trials, new
Health, Family, India, Government, Welfare, Ministry, Government of india, Ministry of health and family welfare
CDSCO - Guidance for Industry
www.cdsco.nic.inGuidance for Industry Central Drugs Standard Control Organization Page 3 OBJECTIVE This Guidance has been developed in conformity with Drugs and Cosmetics
No.DCG (I/Misc./2017 (68) Directorate General of …
www.cdsco.nic.inPage 1 of 1 No.DCG (I/Misc./2017 (68) Central Drugs Standard Control Organisation Directorate General of Health Services Office of Drugs Controller General India
Central Drugs Standard Control Organization
www.cdsco.nic.inCentral Drugs Standard Control Organization Directorate General of Health Services, Ministry of Health and Family Welfare, Government of India
Guidelines on Similar Biologic: Regulatory …
www.cdsco.nic.inGuidelines on Similar Biologic: Regulatory Requirements for Marketing Authorization in India
Guidelines, Requirements, Regulatory, Marketing, Authorization, Biologics, Regulatory requirements for marketing authorization
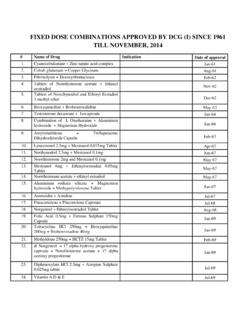
FIXED DOSE COMBINATIONS APPROVED BY DCG …
www.cdsco.nic.inFIXED DOSE COMBINATIONS APPROVED BY DCG (I) SINCE 1961 TILL NOVEMBER, 2014 # Name of Drug Indication Date of approval …
Dose, Till, Combination, Approved, Fixed, Fixed dose combinations approved by dcg
DRAFT GUIDANCE ON APPROVAL OF CLINICAL …
www.cdsco.nic.inguidelines on approval of clinical trial & new drugs effective date: page 1 of 71 draft guidance on approval of clinical trials & new drugs draft guidance
Drug, Guidance, Clinical, Trail, Approval, Guidance on approval of clinical, New drugs, Guidance on approval of clinical trials amp new drugs
CENTRAL DRUGS STANDARD CONTROL …
www.cdsco.nic.incentral drugs standard control organisation directorate general of health services, ministry of health and family welfare government of india
General, Standards, Control, Drug, Central, Organisation, Directorate, Central drugs standard control, Central drugs standard control organisation directorate general
DRAFT GUIDANCE FOR INDUSTRY ON REPORTING …
www.cdsco.nic.indraft guidelines on reporting serious adverse events dated:11-05-2011 page 1 of 9 guidelines on reporting serious adverse event draft guidance for industry on reporting serious adverse events
Draft, Guidance, Reporting, Industry, Draft guidance for industry on reporting
Related documents
COMPANY PROFILE - Hardy Diagnostics …
hardydiagnostics.comPlated Media Our Hardy Products Bottled Media Tubed Media Slide Stainers Hardy Diagnostics manufactures culture media and rapid identification kits for microbiological testing in clinical, research, food, and pharmaceutical laboratories.
Company, Pharmaceutical, Laboratories, Profile, Company profile, Pharmaceutical laboratories
Exclusion of Objectionable Microorganisms from …
www.biomerieux.com.auPresentation Overview •Introduction to the PDA Technical Report on the exclusion of objectionable microorganisms from non-sterile drug products. •Regulatory definition of objectionable microorganisms. •The role of microbial testing in detecting objectionable microorganisms •Decision trees for objectionable organisms in different dosage forms
Baird Parker Agar Base - HiMedia Leading …
www.himedialabs.comHiMedia Laboratories Technical Data Please refer disclaimer Overleaf. Tryptone, HM peptone B and yeast extract are sources of nitrogen, carbon, sulphur and vitamins.
Laboratories, Agar, Baird parker agar base, Baird, Parker, Base
Hazardous Laboratory Waste Disposal - IAP-AD
www.iap-ad.orgSources of Waste • 1) Medical , Veterinary and Agricultural laboratories, Mortuaries Forensic or Educational . • 2) X-ray imaging and Laboratories of photography & film development. • 3) Laboratories of educational institutes. • 4) Printing industry. • 5) Industrial laboratories.(tanning, batteries, fertilizers, perfumes and cosmetics, food, …
Editor: Edgard Delvin Ph.D Centre de Recherche
www.ifcc.orgIFCC eNews May-June 2011 issue chemistry/chemical, medicine/medical, and pharmacy/pharmaceutical in the classification of the different specialties of the clinical laboratory sciences
Annex 2 WHO good practices for pharmaceutical …
www.who.int71 Introduction and scope of document Pharmaceutical microbiology laboratories may be involved in: — sterility testing; — detection, isolation, enumeration and identifi cation of microorganisms
Microbiology, Pharmaceutical, Laboratories, Pharmaceutical microbiology laboratories