U. S. Department of Justice Drug Enforcement Administration
The Controlled Substances Act (CSA), 21 U.S.C. 801 et seq., states that a pharmacist may not dispense a schedule II controlled substance without a written prescription of a practitioner, “except that in emergency situations… such drug may be dispensed upon oral prescription….” 21 U.S.C. 829(a).
Download U. S. Department of Justice Drug Enforcement Administration
Information
Domain:
Source:
Link to this page:
Please notify us if you found a problem with this document:
Documents from same domain
Controlled Substances Act - Justice
www.deadiversion.usdoj.govDrug Enforcement Administration . Pharmacist’s Manual
BENZODIAZEPINES (Street Names: Benzos, …
www.deadiversion.usdoj.govDrug Enforcement Administration Office of Diversion Control Drug & Chemical Evaluation Section BENZODIAZEPINES (Street …
Administration, Enforcement, Drug, Drug enforcement administration
Controlled Substances Act - deadiversion.usdoj.gov
www.deadiversion.usdoj.govUnited States Department of Justice Drug Enforcement Administration Office of Diversion Control . P. harmacist’s . M. anual. An Informational Outline of the
Department of Justice - deadiversion.usdoj.gov
www.deadiversion.usdoj.govVol. 79 Tuesday, No. 174 September 9, 2014 Part II Department of Justice Drug Enforcement Administration 21 CFR Parts 1300, 1301, 1304, et al.
Controlled Substances by DEA Drug Code Number
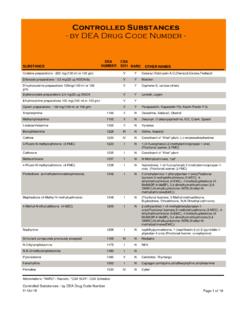
www.deadiversion.usdoj.govControlled Substances - by DEA Drug Code Number - DEA CSA SUBSTANCE NUMBER SCH NARC OTHER NAMES Codeine preparations - 200 mg/(100 ml or 100 gm) V Y Cosanyl,Robitussin A-C,Cheracol,Cerose,Pediacof
Drug, Substance, Controlled, Controlled substances by dea drug
Scheduling Actions Controlled Substances …
www.deadiversion.usdoj.govScheduling Actions - Alphabetical Order - FINAL ORDER SUBSTANCE PROPOSAL FEDERAL *Scheduled under 21 USC 811(h) PUBLICATION PUBLICATION REGISTER EFFECTIVE CSA
Scheduling, Action, Substance, Controlled, Scheduling actions controlled substances
APPLICATION FOR REGISTRATION APPROVED OMB …
www.deadiversion.usdoj.govForm-225 APPLICATION FOR REGISTRATION Under the Controlled Substances Act APPROVED OMB NO 1117-0012 FORM DEA-225 (04-12) FORM EXPIRES: 7/31/2018 INSTRUCTIONS Save time - apply on-line at www.deadiversion.usdoj.gov DEA OFFICIAL …
Form, Applications, Under, Substance, Controlled, Usdoj, Under the controlled substances act, Deadiversion
An Informational Outline of the Controlled …
www.deadiversion.usdoj.govThis manual has been prepared by the Drug Enforcement Administration, Office of Diversion Control, to assist practitioners (physicians, dentists, veterinarians, and other
Outline, Informational, Controlled, Informational outline of the controlled
U. S. DEPARTMENT OF JUSTICE – DRUG …
www.deadiversion.usdoj.govForm DEA-41 See instructions on reverse (page 2) of form. OMB APPROVAL NO. 1117-0007 Expiration Date 10/31/2020 U. S. DEPARTMENT OF JUSTICE – DRUG ENFORCEMENT ADMINISTRATION
Mid-Level Practitioners Authorization by State
www.deadiversion.usdoj.govMid-Level Practitioners Authorization by State Pursuant to Title 21, Code of Federal Regulations, Section 1300.01(b28), the term mid-level practitioner means an individual
States, Levels, Practitioner, Authorization, Mid level practitioners authorization by state
Related documents
How to Prescribe Controlled Substances to Patients During ...
www.deadiversion.usdoj.govthe usual course of his/her professional practice. 21 CFR 1306.04(a). In all circumstances when prescribing a controlled substance, including those summarized below, the practitioner must use his/her sound judgment to determine that s/he has sufficient information to conclude that the issuance of the prescription is for a bona fide medical purpose.
Pennsylvania Code
www.health.pa.govJan 04, 1984 · CHAPTER 25 CONTROLLED SUBSTANCES, DRUGS, DEVICES, AND COSMETICS GENERAL PROVISIONS § 25.1. Definitions. The following words and terms when used in this chapter, have the following meanings, unless the context clearly indicates otherwise: Act—The Controlled Substance, Drug, Device and Cosmetic Act (35 P. S. § § 780-101— 780 …
DEA Registration Requirements for Physician’s Assistants ...
www.michigan.govState of Michigan, mid-level practitioners cannot independently issue a controlled substance prescription without the delegating prescriber’s identity appearing on the prescription. Michigan Board of Pharmacy Administrative Rule 338.3161 requires that a prescription issued for a controlled substance shall include the prescriber’s DEA
Prescription, Michigan, Substance, Controlled, Controlled substances, Controlled substance prescription
Chapter 45H Controlled Dangerous Substances
www.njconsumeraffairs.gov“Controlled dangerous substance” or “controlled substance” means a controlled dangerous substance as defined in N.J.S.A. 24:21-2. “Days” means calendar days. “Director” means the Director of the Division of Consumer Affairs in the Department of Law and Public Safety.
Chapter, Dangerous, Substance, Controlled, Chapter 45h controlled dangerous substances, Controlled substances
2022 LAWBOOK FOR PHARMACY (Business and Professions …
www.pharmacy.ca.govWithout Prescription: Exc eptions . 4059.5. Who May Order Dangerous Drugs or Devices: Exceptions; Compliance with Laws of All Involved Jurisdictions . 4060. Controlled Substance - Prescription Required; Exceptions . 4061. Distribution of Drug as Sample; Written Request Required . 4062. Furnishing Dangerous Drugs during Emergency . 4063.
Prescription, Substance, Controlled, Controlled substance prescription
DEA Information on Telemedicine - Substance Abuse and ...
www.samhsa.govthe pharmacy, or by calling in a schedule III -V prescription to the pharmacy. Important note: If the prescribing practitioner has previously conducted an in-person medical evaluation of the patient, the practitioner may issue a prescription for a controlled substance after having communicated with the patient via telemedicine, or any other
Information, Prescription, Substance, Controlled, Controlled substances, Telemedicine, Dea information on telemedicine