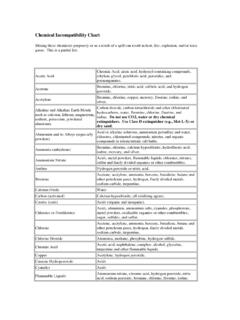
And Alkaline Earth Metals
Found 7 free book(s)Chemical Incompatibility Chart - Princeton University
ehs.princeton.eduAlkaline and Alkaline Earth Metals such as calcium, lithium, magnesium, sodium, potassium, powdered aluminum Carbon dioxide, carbon tetrachloride and other chlorinated hydrocarbons, water, Bromine, chlorine, fluorine, and iodine. Do not use CO2, water or dry chemical extinguishers. Use Class D extinguisher (e.g., Met-L-X) or dry sand.
Periodic Table Electroneg
www.sciencegeek.netAlkali metals Alkaline earth metals Transition metals Lanthanides Actinides Other metals Metalloids (semi-metal) Nonmetals 6.94 Halogens Noble gases Element name 80 Symbol Beryllium Electronegativity Mercury Hg 200.59 1.9 Atomic # Lithium
Periodic Trends Multiple Choice Review PSI Chemistry Name
content.njctl.orgA) Alkali metals B) Transition metals C) Halogens D) Alkaline Earth metals E) Noble gases 39)Which equation correctly represents the first ionization of aluminum? [ ] A) Al-(g) Al(g) +e- B) Al (g) Al-(g) +e- C) Al (g) + e- Al-(g) D) Al (g) Al +(g) +e- E) Al+ (g) +e- Al(g)
Periodic Table and Atomic Structure: Secret Agent
labsci.stanford.edu• 1c. Students know how to use the periodic table to identify alkali metals, alkaline earth metals and transition metals, trends in ionization energy, electronegativity, and the relative sizes of ions and atoms. • 1d. Students know how to use the periodic table to determine the number of electrons available for bonding. • 1e.
GLASS-MAKING MATERIALS. - USGS
pubs.usgs.govmetals. Usually the alkaline bases are sodium and potassium; the alkaline earth is calcium, and the common metallic element is lead. All of these are used'in the form of salts. In melting together the various ingredients employed in the batch or mixture it appears that silica under the influence of heat in the presence of a flux forms silicates ...
Chapter 7
www.lamar.eduThe Alkaline Earth Metals • The chemistry is dominated by the loss of two s electrons: M →→→ M2+ + 2e-. • Be does not react with water. • Mg will only react with steam. • Ca onwards: Ca( s) + 2H 2O( l) →→→ Ca(OH) 2(aq ) + H 2(g)
USGS Mineral Resources Program The Rare-Earth Elements ...
pubs.usgs.govEarth’s crust but rarely in economic concentrations. Economic REE deposits occur primarily in four geologic environments: carbonatites, alkaline igneous systems, ion-absorption clay deposits, and monazite-xenotime-bearing placer deposits. Even within these deposit types, minable (economic) concentrations of REEs are rare.