Assessing The Quality Of Collaboration
Found 7 free book(s)Evidence-Based Practice: What It Is and Why It Matters
www.cdc.govChallenges in assessing evidence in genetics ... Assess quality of evidence Evaluate risk of bias Synthesize results. ... Agency for Healthcare Research & Quality (AHRQ) Cochrane Collaboration Health Technology Assessment International (HTAi) Sources of …
State Law Chart: Nurse Practitioner Practice Authority
www.ama-assn.orgspecify a plan for quality assurance management with established patient ... Scope of practice includes: assessing clients, synthesizing and analyzing data None ... collaboration must address a reasonable and appropriate level of consultation and referral, coverage for the patient in the absence of the APRN, a method to ...
Creating a Continuous Quality Improvement Plan
www.jbassoc.comProcess for monitoring the CQI plan and assessing progress Design Options for Home Visiting Evaluation DOHVE) provides research and evaluation support for the MIECHV Program. DOHVE is funded by the Administration for Children and Families (ACF) in collaboration with the Health Resources and Services Administration (HRSA).
Health Canada
www.canada.caHealth Canada (2020). Guidelines for Canadian Drinking Water Quality—Summary Table. Water and Air Quality Bureau, Healthy Environments and Consumer Safety Branch, Health Canada, Ottawa, Ontario. The document was prepared by Health Canada in collaboration with the Federal-Provincial-Territorial Committee on Drinking Water of the
STRATEGIC PLANNING FOR COMMUNITY DEVELOPMENT
www.library.nd.govhealthy community with a desirable quality of life will require innovative thinking and new attitudes. Solutions will need to be developed through collaborative approaches that involve an expanding leadership base and a more enlightened citizenry. The achievement of effective collaboration can occur using a systematic planning process
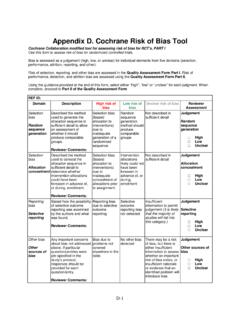
Cochrane Risk of Bias Tool - National Center for ...
www.ncbi.nlm.nih.govCochrane Collaboration modified tool for assessing risk of bias for RCT’s, PART I. Use this form to assess risk of bias for randomized controlled trials. Bias is assessed as a judgement (high, low, or unclear) for individual elements from five domains (selection, performance, attrition, reporting, and …
Laboratory Quality Management System Handbook
www.who.intQuality Management System for all stakeholders in health laboratory processes, from management, to administration, to bench-work laboratorians. This handbook covers topics that are essential for quality management of a public