Exposure Prophylaxis For Hiv
Found 8 free book(s)Recommended ICD-10-CM Codes Pre-Exposure Prophylaxis …
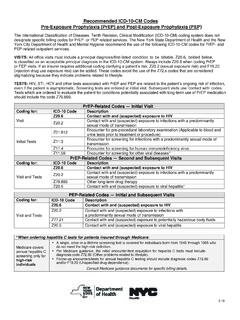
www1.nyc.govZ20.6 Contact with and (suspected) exposure to HIV Z20.2 Contact with and (suspected) exposure to infections with a pr e d o mn ant ly sxu e f tran on Z77.21 Contact with and (suspected) exposure to potentially hazardous body fluids Z20.5 Contact with and (suspected) exposure to viral hepatitis
PREEXPOSURE PROPHYLAXIS FOR THE PREVENTION OF HIV ...
www.cdc.govgovernment patents and patent applications related to methods for HIV prophylaxis. Suggested Citation: Centers for Disease Control and Prevention: US Public Health Service: Preexposure prophylaxis for the prevention of HIV infection in the United States—2021 Update: a clinical practice guideline.
HIV Pre-Exposure Prophylaxis (PrEP): A brief guide for ...
paetc.orgHIV PrEP guidance for providers, updated April 2016 * page 1 of 4 HIV Pre-Exposure Prophylaxis (PrEP): A brief guide for providers updated April 2016 Daily emtricitabine/tenofovir (Truvada®) is safe and effective for reducing the risk of HIV acquisition in sexually active men and women and injection drug users when used consistently.
Alberta HIV Pre-Exposure Prophylaxis (PrEP) Guidelines
www.albertahealthservices.caThe newest addition to the list of existing HIV prevention strategies, HIV pre-exposure prophylaxis (PrEP), when taken daily, is a highly effective pharmacologic means of preventing HIV infection. PrEP should be used as part of a comprehensive HIV prevention strategy involving the care and treatment of HIV infected individuals, condom use, and
HIV pre-exposure prophylaxis (PrEP) 2018
www.bhiva.orgSep 11, 2017 · Melinda Tenant-Flowers Retired Consultant in HIV and Sexual Health Medicine, King’s ollege Hospital, London Ed Wilkins Consultant in Infectious Diseases, North Manchester General Hospital Ingrid Young Chancellor's Fellow, Usher Institute, University of Edinburgh BHIVA/BASHH guidelines on the use of HIV pre-exposure prophylaxis (PrEP) 2018
UK Guideline for the use of HIV Post-Exposure Prophylaxis …
www.bhiva.orgin people using HIV pre-exposure prophylaxis (PrEP) – please to the full guideline. 1. When to offer PEP (section 6): Where the index partner is HIV-positive, has been on antiretroviral therapy for at least 6 months with an undetectable plasma HIV viral load (at the time of last measurement and within the last 6 months) and with
CDC HIV-NPEP Guidelines
www.cdc.govOther Nonoccupational Exposure to HIV— United States, 2016 from the Centers for Disease Control and Prevention, U.S. Department of Health and Human Services . Update: Interim Statement Regarding Potential Fetal Harm from Exposure to Dolutegravir – Implications for HIV Post-exposure Prophylaxis (PEP). Please see attached file.
Guidelines for the Management of Occupational Exposures to ...
www.who.intRisk Factors for HIV Transmission After Percutaneous Exposure to HIV-Infected Blood: Risk Factor Adjusted Odds ratio (95% CI) Deep injury 15 (6.0-41) Visible blood on device 6.2 (2.2-21) Procedure involving needle 4.3 (1.7-12) placed in artery …