Mole Worksheet 2
Found 6 free book(s)CHEMISTRY COMPUTING FORMULA MASS WORKSHEET
www.isd622.orgCHEMISTRY MOLAR RATIOS WORKSHEET 2 1 1 2 2 2 2 3 3 2. The Mathematics of Chemical Equations ... X = 90 g O2 1 mole O2 2 mole KClO3 122.5 g KClO3 = 229.7 g KClO3 32 g O2 3 moles O2 1 mole KClO3 I II III IV CHEMISTRY STOICHIOMETRY Mass (g,kg, etc.) Volume (L, mL. etc.) or # of Items (atoms,molec.) ...
Gas Law's Worksheet - Willamette Leadership Academy
www.willametteleadershipacademy.netCHEMISTRY GAS LAW’S WORKSHEET 20. Determine the molar mass of a gas that has a density of 2.18 g/L at 66°C and 720 mm Hg. (Hint: the number of moles of a substance is its mass/molecular mass and density is mass/volume.) 19. What is the pressure in atm exerted by 2.48 moles of a gas in a 250.0 mL container at 58°C? 18.
Reading Street Stories - 1st Grade
www.clinton.k12.ky.usMole and the Baby Bird Retelling Card Pictures color.doc Dot and Jabber Retelling Pictures color.pdf Simple Machines Retelling Pictures color.pdf Alexander Graham Bell Retelling Pictures.pdf Ben Franklin and His First Kite Retelling Cards color.pdf Retelling Center.pdf Green Group Retelling Cards Center Worksheet.pdf
WORKSHEET-Born-Haber Cycle - Cerritos College
www.cerritos.eduWORKSHEET-Born-Haber Cycle 1. a. Draw Born-Haber cycle for the formation of strontium chloride ... The enthalpy of sublimation of strontium = + 164 kj/mole First ionization energy for strontium = + 549 kj/mole Second ionization energy for strontium = + 1064 kj/mole The enthalpy of dissociation of chlorine, Cl2 = + 243 kj/mole ... 2. Name the ...
AQA, OCR, Edexcel GCSE Science - MME
mathsmadeeasy.co.uk2 + Al2(SO 4) 3 BaSO 4 + AlCl 3 (4 marks) Limiting reactants Q10: Describe what is meant by the term ‘a limiting reactant’. (3 marks) Q11: In the following equation: Zn + I 2 ZnI 2 If 3 moles of zinc are used and only 1 mole of iodine, what is the mass of the product, zinc iodide? (2 marks)
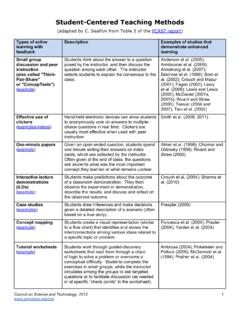
Student-Centered Teaching Methods
teachinghandbook.wayne.eduheld models and computer imaging programs helps students answer oral questions about mole cular structure and function: A controlled investigation of student learning.” CBE—Life Sciences Education 8(1): 29-43. Heller, P., R. Keith, and S. Anderson. (1992). "Teaching Problem Solving Through Cooperative Grouping.