Multiple reactions
Found 8 free book(s)Some Reactions of Hydrocarbons
myweb.liu.eduReactions of Hydrocarbons Experiment #2 Pre-Lab Exercise 1. How would you describe the difference between saturated and unsaturated hydrocarbons and what is a distinguishing feature of aromatic hydrocarbons. 2. Describe the relative reactivity of alkanes, alkenes and aromatic hydrocarbons with respect to their chemical reactions.
Multiple Choice Questions
www.ncert.nic.inMultiple Choice Questions 1. Which of the following correctly represents 360 g of water? (i) 2 moles of H 2 0 ... number of water molecules through a complex series of reactions to give a molecule of glucose having a molecular formula C 6 H 12 O 6. How many grams of water would be required to produce 18 g of glucose? Compute the volume
Understanding Your Multiple Myeloma Lab Tests
www.velcade.comIn multiple myeloma, one plasma cell goes bad and makes multiple copies of itself (clones). Each copy makes the same antibody protein or M spike. SPEP is used to separate and identify the presence and levels of M protein in the blood. Each type of plasma cell produces only 1 type of immunoglobulin. Beta2-microglobulin (B2M), serum 8
Guideline on good pharmacovigilance practices (GVP)
www.ema.europa.eumanagement and reporting of suspected adverse reactions (serious and non-serious) associated with medicinal products for human use authorised in the European Union ( EU). Recommendations regarding the reporting of emerging safety issues or o f suspected adverse reactions occurring in special situations are also presented in this Module.
Understanding your MULTIPLE MYELOMA LAB TESTS
www.ninlarohcp.comorgans (kidney, liver, etc) that multiple myeloma and chemotherapy may affect. Glucose, serum Normal range: 70-100 mg/dL when fasting Blood sugar, or glucose, is the main sugar found in your blood. Your blood carries glucose to all of your body’s cells to use for energy. Blood urea nitrogen (BUN) Normal range: 7-20 mg/dL
Second Order Linear Differential Equations
www.personal.psu.edut is a solution, and so is any constant multiple of it, C1 e t. Not as obvious, but still easy to see, is that y 2 = e −t is another solution (and so is any function of the form C2 e −t). It can be easily verified that any function of the form y = C1 e t + C 2 e −t will satisfy the equation. In fact, this is the general solution of the
Adult Immunization Schedule - AAFP Home
www.aafp.orgReport all clinically significant postvaccination reactions to the Vaccine Adverse Event Reporting System (VAERS). Reporting forms and instructions on …
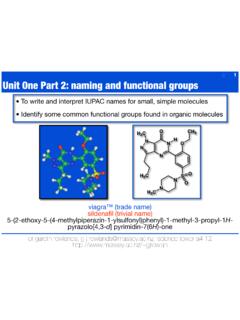
Unit One Part 2: naming and functional groups
www.massey.ac.nz• Saturated hydrocarbons - contain only C & H and no multiple bonds • Non-cyclic alkanes have the formula CnH2n+2 8 H C H H H methane CH4 propane C3H8 pentane C5H12 cyclopentane C5H10 • Alkanes are a little dull - used as solvents or burnt as fuel • Methane is a fuel, a chemical feedstock & a green house gas propane CH4