Of Reaction Kinetics For Chemical Reaction Engineering
Found 13 free book(s)Fundamentals of Chemical Reactor Theory1
www.seas.ucla.eduChemical kinetics and reactor engineering are the scientific foundation for the analysis of most environmental engineering processes, both occurring in nature and invented by men. The need to quantify and compare processes led scientists and engineers throughout last century to develop what is now referred as Chemical Reaction Engineering (CRE).
Continuous Stirred Tank Reactors (CSTRs)
ocw.mit.edu1st order reaction kinetics C C A0 A1 = 1+Da1 For the second reactorÆ iterate ()( ) C C A0 A2 = 11++Da12Da If the CSTRs are identical, () C CAn = A0 1+Da n Æ many CSTRs in series looks like a plug flow reactor. 10.37 Chemical and Biological Reaction Engineering, Spring 2007 Lecture #5 Prof. William H. Green Page 4 of 4
Biological Reactors (Chemostat) - MIT OpenCourseWare
ocw.mit.eduogistic equation” “L x t Figure 5. Graph of logistic growth. If product is something cells are making: Product synthesis kinetics 1) 10.37 Chemical and Biological Reaction Engineering, Spring 2007 Lecture 13
Chemical Engineering Kinetics - Tufts University
sites.tufts.eduwhich is typical rate law kinetics. In this expression, if , , ,… are the stoichiometric coefficients of a reaction, it may be (but is not necessarily) an elementary step. Typically, we find that for elementary steps ∑ …<3. 2.2.2 FIRST ORDER KINETICS We will start by considering the elementary reaction →
Design and Control of a Methanol Reactor/Column Process
terpconnect.umd.eduJun 02, 2010 · The reaction rate for the water-shift reaction is given in eq 5. Table 1 gives the kinetic and adsorption parameters entered into the Aspen LHHW reaction model to implement these kinetics. Since the reactions are exothermic, the chemical equilibrium constants decrease with increasing temperature. Therefore, low
Basic Principles and Calculations in Chemical Engineering
ptgmedia.pearsoncmg.comview the study of the field of chemical engineering as a tree with material and energy balances being the trunk and the subjects of thermodynamics, fluid flow, heat transfer, mass transfer, reactor kinetics, process control, and
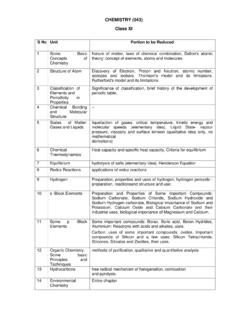
CHEMISTRY (043) Class XI
cbseacademic.nic.inB. Chemical Kinetics a. Effect of concentration and temperature on the rate of reaction between Sodium Thiosulphate and Hydrochloric acid. b. Study of reaction rates of any one of the following: i) Reaction of Iodide ion with Hydrogen Peroxide at room temperature using different concentration of Iodideions.
Mathematical Modelling in Systems Biology: An Introduction
www.math.uwaterloo.cachemical kinetics, providing rate laws for biochemical processes (i.e. enzyme-catalysed reactions and cooperative binding). An optional section treats common approximation methods. Chapter 4 introduces techniques for analysis of differential equation models, including phase plane analysis, stability, bifurcations, and sensitivity analysis.
Production of Gold - Chemical and Biomedical Engineering ...
cbe.statler.wvu.eduhigh enough so oxygen transfer was limiting). For this reaction, a resistance in series expression can be developed and then used in a first order rate of reaction expression developed by Habashi 3. The adsorption of gold from the pregnant leach solution onto activated carbon is a key step in the recovery process.
CHAPTER 4:The Material Balance for Chemical Reactors
sites.engineering.ucsb.eduThis is the subject of chemical kinetics, Chapter 5 Here we use common reaction-rate expressions without derivation 4/152. The Batch Reactor R j The batch reactor is assumed well stirred Let the entire reactor contents be the reactor volume element 5/152
Get help and support AS AND A-LEVEL CHEMISTRY
filestore.aqa.org.uk3.1.5 Kinetics page 20 3.1.6 Chemical equilibria, Le Chatelier’s principle and K c page 22 3.1.7 Oxidation, reduction and redox equations page 24 3.1.8 Thermodynamics (A-level only) page 25 3.1.9 Rate equations (A-level only) page 27 3.1.10 Equilibrium constant K p for homogeneous systems (A-level only) page 29
Hydrogen production by electrolysis - .NET Framework
energiforskmedia.blob.core.windows.netAnode reaction 2Cl--→ Cl. 2 + 2e. Chlor-alkali . 2Cl-+ 2H. 2. O → -Cl. 2 + H. 2 + 2OH ~60 million tonnes/year Cl. 2 . produced world wide . Many uses for the products . Chlorate . NaCl + 3H 2 O → NaClO 3 + 3H 2 . About 4 million tonnes/year NaClO 3 produced world wide . Main use in the bleaching of chemical pulp . In some plants the ...
UNIVERSITAT DE BARCELONA REACTORES MULTIFÁSICOS
diposit.ub.eduuniversitat de barcelona 2013 reactores multifÁsicos apuntes montserrat iborra. javier tejero. fidel cunill.