Search results with tag "Seasonal influenza"
2018/19 Seasonal Influenza Vaccine Eligibility
www.bccdc.ca2018/19 Seasonal Influenza Vaccine Eligibility . Trivalent and Quadrivalent Inactivated Influenza Vaccines (TIIV & QIIV), and Quadrivalent Live Attenuated Influenza Vaccine (LAIV-Q)
This is an official CDC HEALTH ADVISORY
emergency.cdc.govseveral states. Influenza vaccination coverage is still low and there is still time this season to benefit from getting an annual influenza vaccine. Available seasonal influenza vaccines in the United States provide protection against four different influenza viruses: A(H1N1)pdm09, A(H3N2), B/Victoria lineage, and B/Yamagata lineage viruses. In the
WHO information for the molecular detection of influenza ...
www.who.int6 Referral for further characterization Human specimens with laboratory results indicative of influenza A that are unsubtypeable (i.e. negative for seasonal influenza A(H1), A(H3)) or detected/diagnosed as non-seasonal HA
The elusive definition of pandemic influenza
www.who.intseasonal influenza required different management, a premise reinforced by the decision on the part of the World Health Organization (WHO) to label the H1N1 influenza outbreak a “pandemic”. However, the outbreak had far less serious con-sequences than experts had predicted, a fact that led many to
Data at a Glance February 27th March 5th 2022 (Week 9)
www.oregon.govUS Data (from CDC FluView): Seasonal influenza activity in the United States remains low, but the num-ber of influenza virus detections reported by public health laboratories has increased in recent weeks. Viruses • Clinical Labs: The percentage of respiratory specimens testing positive for influenza at clinical labora-tories is 5.8% this week.
VACCINATION AND IMMUNISATION PROGRAMMES 2015/16
www.nhsemployers.orgV&I Guidance July 2015 2 Version control Version Publication date Changes Version 1.1 26-03-2015 This version includes: Childhood seasonal influenza Hepatitis B HPV booster Measles mumps rubella MenC booster MenC freshers Pertussis Rotavirus Seasonal influenza and pneumococcal polysaccharide
ACIP--FDA-CBER Plans for Monitoring COVID-19 Vaccine ...
www.cdc.govOct 30, 2020 · Near real-time, rapid surveillance in 2017 -2018 seasonal influenza vaccine – evaluation of 6 health outcomes of interest. 18. ... effectiveness studies for influenza and zoster vaccines • Conducted duration of effectiveness analysis of Zostavax vaccine. 21. 4. US Government-wide Efforts
HEALTH CARE PROVIDER INFLUENZA VACCINE CONSENT …
www.healthunit.comI consent to receiving the seasonal influenza vaccine. If signing for someone other than yourself, indicate your relationship to that other person: _____ If signing for someone other than myself, I confirm that I am the parent / legal guardian or substitute decision maker.
Keep Flu out of School
www.preventchildhoodinfluenza.orgOverview of Seasonal Influenza (Flu) Influenza, also called flu, is among the leading causes of preventable morbidity and mortality. In fact, flu causes more deaths than any other vaccine-preventable disease
Fillable Vaccine Administration Consent Form
www.centuryrx.netSeasonal Influenza COVID-19 Hepatitis A Hepatitis B Chickenpox (varicella) HPV Pneumococcal Tetanus/TDap Shingles (zoster) Meningococcal MMR Other Vaccine Administration Consent Form Section A (Please print clearly.) First name: Last name:
Health Care Professional and Provider Manual
www.selecthealthofsc.comReimbursement Policy: Molecular Diagnostic Infectious Disease Testing (including seasonal influenza testing) . . 67 Reimbursement Policy: Presumptive and Definitive Drug Testing .
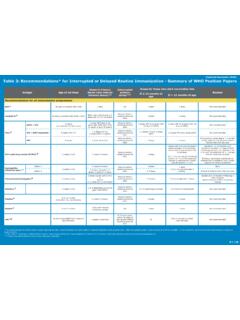
Table 3: Recommendations* for Interrupted or Delayed ...
www.who.intSeasonal influenza (inactivated tri- and qudri-valent) ≥ 9 yrs: 1 dose21 6 months (min) < 9 yrs: 2 doses (4 weeks) Resume without repeating previous dose 2 doses < 9 yrs: 2 doses ≥ 9 yrs: 1 dose Revaccinate annually 1 dose only Varicella 22 12-18 months 1-2 (4 weeks – 3 months, depending on manufacturer) Resume without repeating previous dose
Seasonal Influenza Vaccine Production Process
www.vaccineplace.comaTo ensure safety and purity, vaccine is produced in a clean environment where quality control experts enforce strict standards, continuously monitoring the process; bThe