Guideline on Live Recombinant Viral Vectored Vaccines
Although vaccines containing live-attenuated viruses are typically not adjuvanted, guidance on the co-use of immunomodulators is provided by the guideline on Adjuvants in Vaccines for Human Use (CHMP/VEG/134716/04)[3] and by the accompanying Explanatory Note (CHMP/VWP/244894/2006)[4]. 4. Legal basis
Tags:
Live, Recombinant, Vaccine, Viral, Adjuvanted, Vectored, Live recombinant viral vectored vaccines
Information
Domain:
Source:
Link to this page:
Please notify us if you found a problem with this document:
Documents from same domain
Q12 Step 2b Technical and regulatory …
www.ema.europa.euICH guideline Q12 on technical and regulatory considerations for pharmaceutical product lifecycle management EMA/CHMP/ICH/804273/2017 Page 3/36
Product, Management, Lifecycle, Product lifecycle management
Guideline on good pharmacovigilance practices …
www.ema.europa.eu9 December 2013 . EMA/816292/2011 Rev 1* Guideline on good pharmacovigilance practices (GVP) Module VII – Periodic safety update report (Rev 1)
Guidelines, Good, Practices, Guideline on good pharmacovigilance practices, Pharmacovigilance
Guideline on good pharmacovigilance practices (GVP)
www.ema.europa.euGuideline on good pharmacovigilance practices (GVP) – Module IX (Rev 1) EMA/827661/2011 Rev 1 Page 2/25
Guideline on good pharmacovigilance practices …
www.ema.europa.euGuideline on good pharmacovigilance practices (GVP) – Module VIII (Rev 3) EMA/813938/2011 Rev 3 Page 2/28
Guidelines, Good, Practices, Guideline on good pharmacovigilance practices, Pharmacovigilance
Guideline for good clinical practice E6(R2)
www.ema.europa.euGuideline for good clinical practice E6(R2) EMA/CHMP/ICH/135/1995 Page 2/70 Document History First Codification History Date New Codification
Guideline for good clinical practice E6(R2)
www.ema.europa.euGuideline for good clinical practice E6(R2) EMA/CHMP/ICH/135/1995 Page 2/75 10 Document History 11 First Codification History Date New Codification November
Guidelines, Good, Practices, Clinical, Good clinical practice e6
Guideline on good pharmacovigilance practices …
www.ema.europa.euharmful physical or psychological effects [DIR 2001/83/EC Art 1(1 6)]. 74 Adverse event (AE); synonym: Adverse experience 75 Any untoward medical occurrence in a patient or clinical- trial subject administered a medicinal product
Guidelines, Good, Practices, Guideline on good pharmacovigilance practices, Pharmacovigilance
European Medicines Agency
www.ema.europa.eu© EMEA 2006 2 SPECIFICATIONS: TEST PROCEDURES AND ACCEPTANCE CRITERIA FOR BIOTECHNOLOGICAL/BIOLOGICAL PRODUCTS ICH Harmonised Tripartite Guideline
Guidelines, European, Agency, Medicine, Harmonised, European medicines agency, Ich harmonised
products1/traditional herbal medicinal products
www.ema.europa.euThere is no expectation that existing herbal medicinal products on the market will be affected by this guideline, with the exception of traditional herbal medicinal products for human use that were already
Product, Medicinal, Traditional, Herbal, Products1 traditional herbal medicinal products, Products1
Q7 Q&A - good manufacturing practice for active ...
www.ema.europa.euICH guideline Q7 on good manufacturing practice for active pharmaceutical ingredients – questions and answers EMA/CHMP/ICH/468930/2015 Page 2/37
Good, Practices, Pharmaceutical, Manufacturing, Active, Ingredients, Good manufacturing practice for active, Good manufacturing practice for active pharmaceutical ingredients
Related documents
Module 2.6: Nonclinical Written and Tabulated Summary 2.6 ...
www.mhlw.go.jpGSK™s AS03-adjuvanted H5N1 influenza vaccine is a monovalent, split-virion, inactivated, adjuvanted vaccine. The influenza strain contained in the vaccine is A/H5N1/Indonesia/5/2005, prepared by reverse genetics. The antigen is presented in a 10mL Type I glass vial (10 doses) and the AS03 adjuvant presented in a 3mL Type I glass vial (10 doses).
Evidence Assessment: Sinopharm/BBIBP COVID-19 vaccine
cdn.who.intApr 02, 2021 · • 2-dose β-propiolactone-inactivated, aluminiumhydroxide-adjuvanted COVID-19 vaccine administered on a 0/21-28-day schedule for the prevention of COVID-19 disease • Authorized by the China National Medical Products Administration on December 31, 2020 • Authorized by 45 countries/jurisdictions for use in adults ≥18 years
COVID-19 Clinical Working Group Summary Document Topic ...
media.tghn.orgmay contribute more to the efficacy of adenoviral and mRNA than protein-based vaccines, including adjuvanted subunit and nanoparticle vaccines. The complex, composite nature of protection, as captured in the equation, is further illustrated by (i) the early onset of protection against symptomatic COVID-19 (i.e., 10 days after the first
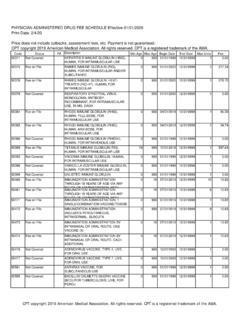
PHYSICIAN ADMINISTERED DRUG FEE SCHEDULE Effective …
www.medicaid.ms.govadjuvanted preservative free 0.5 mg dosage for intramuscular use 0 999: 01/01/2020 12/31/9999: 1 0.00: 90698 fee on file: diphtheria, tetanus toxoids, acellular pertussis vaccine, haemophilus influenzae type b, and inactivated poliovirus vaccine, for intramuscular use 0 18: 08/01/2008 12/31/9999: 1 0.00: 90700 fee on file: diphtheria, tetanus ...