More Practice: Energy, Frequency, Wavelength and the ...
A photon with enough energy, 5.1 electron volts (eV) of energy - to be precise, will eject an electron from a piece of gold! What frequency and wavelength does light with this energy have? Note: 1eV = 1.60 × 10-19 joules !=!
Tags:
Information
Domain:
Source:
Link to this page:
Please notify us if you found a problem with this document:
Documents from same domain
Thermodynamics Unit Internal Energy, Work and Heat
ch301.cm.utexas.edu3. The change in internal energy for the combustion of 1.00 mol CH 4(g) in a cylinder according to the reaction CH4(g) + 2 O2(g) CO2 (g) + 2 H20(g) is ‐892.4 kJ. If a piston connected to the cylinder performs 492 kJ of expansion work due to the combustion,
Kinetic Molecular Theory Supplemental Worksheet
ch301.cm.utexas.eduKinetic Molecular Theory – Supplemental Worksheet 1. What assumptions do we make when using the ideal gas laws? Are the assumptions always true? 2. Consider 2 gases, A and B, each in a 1.0 L container with both gases at the same temperature and pressure. ... mole fraction of methane in the mixture is 0.623. Calculate the total kinetic energy ...
Worksheet, Laws, Theory, Supplemental, Moles, Molecular, Kinetics, Gas law, Kinetic molecular theory supplemental worksheet
Ionic and Covalent Compounds Name: KEY
ch301.cm.utexas.eduIonic and Covalent Compounds Name: KEY!! 1. We differentiate between two types of compounds: IONIC and COVALENT. ... C12H22O11 is MOLECULAR or COVALENT compound, while sodium chloride (table salt) is _ an IONIC compound. ! 6. Carbon monoxide, CO, is an example of a diatomic molecule, while ammonia and glucose, NH3 and C6H12O6, are examples of ...
Name, Compound, Covalent, Ionic, Covalent compound, Ionic and covalent compounds name
IonicBonding!and!Writing!Formulas!Name:!KEY!!! Part A. Use ...
ch301.cm.utexas.edu4. SO3 sulfur trioxide ! 5. N 2O dinitrogen monoxide ! 6. NO nitrogen monoxide ! 7. N2O3 dinitrogen trioxide ! 8. NO2 nitrogen dioxide ! 9. 19.N2O4 2 dinitrogen tetroxide ! 10. N2O5 dinitrogen pentoxide 11. PCl3 phosphorous trichloride ! 12. PCl5 phosphorous pentachloride ! 13. NH3nitrogen trihydride ! 14. SCl6sulfur hexachloride ! 15 ...
University of Texas at Austin
ch301.cm.utexas.eduCOVALENT BONDING Name Covalent bonding occurs when two or more nonmetals share electrons. attempting to attain a stable octet of electrons at least part of the time. For example: Note that hydrogen Is content with 2, not 8. electrons. Show how covalent bonding occurs in each of the following pairs of atoms. Atoms may
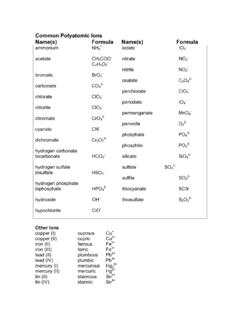
Common Polyatomic Ions Name(s) Formula Name(s) Formula
ch301.cm.utexas.eduCommon Polyatomic Ions Name(s) Formula Name(s) Formula ammonium NH4 + acetate CH3COO-C2H3O2-bromate BrO3-carbonate CO3 2-chlorate ClO3-chlorite ClO2-chromate CrO4 2-cyanide CN-dichromate Cr2O7 2-hydrogen carbonate bicarbonate HCO3-hydrogen sulfate bisulfate HSO4-hydrogen phosphate biphosphate HPO4 2-hydroxide OH-
Kinetics formulas copy - Chemistry 301
ch301.cm.utexas.eduHalf-life (t 1/2): The time it takes for the concentration to drop to one half its current value during the course of the reaction. Note that the “current value” is typically the initial starting value - but not always. Rate Laws for: a A products (all the following equations assume that k is for the overall reaction)
LEWIS STRUCTURES PRACTICE WORKSHEET
ch301.cm.utexas.eduLEWIS STRUCTURES PRACTICE WORKSHEET Draw the Lewis Structures for each of the following molecules. If you are not sure if your structure is correct, do a formal charge check. You should consult the Lewis structure rules and a periodic table while doing this exercise. A periodic table will be available for the exam, but the list of rules will ...
Related documents
Production of X-rays - Western Kentucky University
www.wku.eduPhoton Energy and Power (cont’d) •However, an X-ray beam consists of a spectrum (a distribution) of photon energies, and the rate at which energy is delivered by a beam is determined by the number of photons of each energy. •If there are relatively more low energy photons, it is possible for the low energy component to deliver more energy.
University, Kentucky, Energy, Western, Photon, Western kentucky university, Photon energy
Interactions of Photons with Matter - MIT OpenCourseWare
ocw.mit.eduphoton energy to electron energy. • These electrons will ultimately deposit their energy in the medium. • Photons are far more penetrating than charged particles of similar energy. Energy Loss Mechanisms • photoelectric effect • Compton scattering • pair production Interaction probability • linear attenuation coefficient, µ,
Energy, Mit opencourseware, Opencourseware, Photon, Photon energy
The Compton Effect-- Compton Scattering and Gamma Ray ...
www.phys.utk.eduenergy and momentum are transferred to the charged particle while the photon moves off with a reduced energy and a change of momentum. Generally, the charged particle is an electron considered to be at rest and the photon is usually considered to be an energetic photon such as an X-ray photon or gamma ray photon. In this experiment gamma rays
Compton Scattering - High Energy Physics
pages.hep.wisc.edutered photon is completely correlated with the energy of the scattered photon by energy and momentum conservation. This relation is usually written as: ∆λ = λ0 −λ = h mc (1−cosθ) (1) where λ and λ0 are the wavelengths of the incident and scattered photon respectively, and θ is the photon scattering angle.
Spontaneous and stimulated processes Einstein A and B ...
www.brown.eduConservation of energy: E excited-E ground = E photon "spontaneous emission" - the decay of an excited state to the ground state with the corresponding emission of a photon The two-level atom. Three things can occur •Promotes molecule to a higher energy state •Decreases the number of photons
Attenuation of Radiation - University of Tennessee
www.phys.utk.eduIn pair production, the gamma ray photon’s energy is used to form an electron-positron pair. The rest energy of an electron, and also of a positron, is mc2 where m is the mass of the electron or positron and c is the speed of light. The rest energy of an electron or a positron is 0.511 MeV, so that for an electron-positron pair a minimum ...
6.007 Lecture 39: Schrodinger equation - MIT …
ocw.mit.eduof a photon is proportional to its wavevector k. 2. The energy E of a photon is proportional to its phase velocity v p. 3. We do not experience the wave nature of matter in everyday life because the wavelengths are too small.