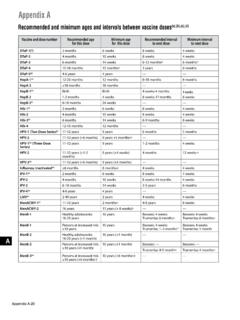
Myocarditis and COVID-19 Vaccine Intervals
Feb 04, 2022 · Within a 7-day risk period after either dose, the rate ratio of myocarditis for Moderna vaccine vs. an unvaccinated comparator was higher than Pfizer vaccine vs. an unvaccinated comparator – The highest rate ratios observed were among those receiving a second dose of Moderna in a heterologous mRNA primary series . VaST:
Tags:
Information
Domain:
Source:
Link to this page:
Please notify us if you found a problem with this document:
Documents from same domain
2018 Combined Recommended Immunization …
www.cdc.govRecommended Immunization Schedule for Children and Adolescents Aged 18 Years or Younger, UNITED STATES, 2018 Approved by the Advisory Committee on …
School Chemistry Laboratory Safety Guide
www.cdc.govSchool Chemistry Laboratory Safety Guide. October 2006. U.S. Consumer Safety Product Commission DEPARTMENT OF HEALTH AND HUMAN SERVICES. Centers for Disease Control and Prevention
Guide, Laboratory, Center, School, Control, School chemistry laboratory safety guide, Chemistry, Safety, Centers for disease control and prevention, Disease, Prevention
Biosafety in Microbiological and Biomedical …
www.cdc.goviii. Foreword. Biosafety in Microbiological and Biomedical Laboratories (BMBL) quickly became the cornerstone of biosafety practice and policy in the United States upon first
Sobering Facts: Drunk Driving in Georgia
www.cdc.govSobering Facts: Drunk Driving in GEORGIA Keep Georgia safe. Keep drunk drivers off the road. This fact sheet provides a snapshot of alcohol-involved deaths and drunk driving
ALCOHOLS III 1402
www.cdc.govALCOHOLS III: METHOD 1402, Issue 2, dated 15 August 1994 - Page 3 of 4 a. Remove and discard back sorbent section of a media blank sampler. b. Inject a known amount of analyte or DE stock solution directly onto front sorbent section
Fast Facts
www.cdc.govAny person working outdoors is at risk of exposure to poisonous plants, such as poison ivy, poison oak, and poison sumac. When in contact with skin, the sap oil (urushiol) of
Fact, Plants, Fast facts, Fast
Trans Fat: The Facts
www.cdc.gov• Read the Nutrition Facts label and ingredient list to compare foods. » Choose products with 0 grams trans fat. » Check the Ingredient List to see if there is any partially hydrogenated oil in the product.
ANTIBIOTIC RESISTANCE THREATS
www.cdc.govApr 23, 2013 · 6. ANTIBIOTIC RESISTANCE THREATS IN THE UNITED STATES, 2013. Executive Summary. Antibiotic Resistance Threats in the United States, 2013. is a snapshot of the complex problem
Chlorine Residual Testing
www.cdc.govChlorine Residual Testing Fact Sheet, CDC SWS Project 4 Methods to Test Free Chlorine in the Field in Developing Countries There are three main methods to test free chlorine residual in drinking water in the field
Testing, Water, Field, Chlorine residual testing, Chlorine, Residual
Traveling? Make Sure You Protect Yourself from …
www.cdc.govCS26201 Malaria transmission occurs throughout Malaria transmission occurs in some parts Malaria transmission is not known to occur Center for Global Health
Related documents
SOPs for reduced interval for 2nd dose for Covishield ...
www.mohfw.gov.in1. Presently, based on the recommendations by National Expert Group on Vaccine Administration for COVID-19 (NÉGVAC), the schedule of Covishield vaccine under National Covid-19 Vaccination Strategy is to administer the 2nd dose at 12-16 weeks interval (i.e. after 84 days), after administration of 1st dose. 2.
COVID-19 – mRNA Pfizer – Ultra Frozen Vaccine Adult 12 ...
www.albertahealthservices.careduced response to vaccines, will vary depending on the immunocompromising condition. Thus, a shortened interval no less than 28 days may be considered for ... them, and as a result received a mixed schedule, it is reasonable to not disadvantage ... vaccine series. • Dose 4 if needed – at least 28 days after the third dose for those with a ...
Ultra, Schedule, Dose, Reduced, Vaccine, Pfizer, Frozen, Rman, 4 dose, Mrna pfizer ultra frozen vaccine
BOOSTRIX (Tetanus Toxoid, Reduced Diphtheria Toxoid and ...
gskpro.comlast dose of the Diphtheria and Tetanus Toxoids and Acellular Pertussis (DTaP) series or 5 years or more after a dose of Tetanus and Diphtheria Toxoids Adsorbed (Td). BOOSTRIX may be administered as an additional dose 9 years or more after the initial dose of Tetanus Toxoid, Reduced Diphtheria Toxoid and Acellular Pertussis Vaccine Adsorbed (Tdap).
Appendix A: Schedule and Recommendations: …
www.cdc.govperiod may be used. If the third dose was administered on or after December 16, 2016, and was administered 12 weeks after the 2nd dose and 5 months after the first dose, it is a valid dose. The 4-day grace period may be used. (o) One dose of influenza vaccine per season is recommended for most persons.
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS
www.ema.europa.euOne single dose of 0.5 ml. Prevenar 13 vaccine schedule for infants and children previously vaccinated with Prevenar (7-valent) (Streptococcus pneumoniae serotypes 4, 6B, 9V, 14, 18C, 19F, and 23F) Prevenar 13 contains the same 7 serotypes included in Prevenar, using the same carrier protein CRM197.
Meningococcal vaccines for Australians - NCIRS
ncirs.org.audemonstrated the immunogenicity of MenACWY vaccine in children, adolescents and adults. All studies indicate that MenACWY vaccines are safe and immunogenic. 22-24 It is preferable to use the same brand of MenACWY vaccine to complete a primary vaccination course. Any brand of vaccine may be used as a booster dose.
California Code of Regulations Title 17, Division 1, Chapter 4
eziz.orgEffective July 1, 2019 Page 1 of 13 IMM-1080 (9-18) California Code of Regulations Title 17, Division 1, Chapter 4 Subchapter 8. Immunization Against Poliomyelitis, Diphtheria, Pertussis, Tetanus,
Code, Title, Regulations, California, California code of regulations title