Chapter 24 Chemistry of Coordination Compounds
Chapter 24! Chemistry of Coordination Compounds! Coordination Compounds! • How do we think about transition metals binding to other atoms? • What do those d orbitals do? • We call them, coordination compounds. Complexes! ... Chapter 9, end and Chapter 24. 4--
Download Chapter 24 Chemistry of Coordination Compounds
Information
Domain:
Source:
Link to this page:
Please notify us if you found a problem with this document:
Documents from same domain
Standard Operating Procedure: Pumps
www2.chemistry.msu.eduStandard Operating Procedure: Pumps Last Updated: ... operation of ports and evacuation procedures are covered in the glove box SOP document. As a general
Operating, Standards, Pumps, Procedures, Standard operating procedure
Radiation Detection & Measurement Dept. of …
www2.chemistry.msu.eduRadiation Detection & Measurement Dept. of Chemistry --- Michigan State Univ. National Superconducting Cyclotron Lab DJMorrissey Spring/2oo9
Chemistry 151 - Michigan State University
www2.chemistry.msu.eduChemistry 151 • Professor James H. Geiger • Office: Chemistry Building, Room 9 ... Chemistry, the Central Science, 10th edition, Prentice-Hall, 2005. ISBN: 0-13-109686-9. ... Registering for Mastering Chemistry, What You Need: A valid email address
Chemistry, Sciences, Central, The central science, Chemistry 151
Chapter 3 Stoichiometry - Michigan State University
www2.chemistry.msu.eduChapter 3! Stoichiometry: Calculations with Chemical Formulas and Equations. Stoichiometry Anatomy of a Chemical Equation CH 4 (g) + 2O 2 (g) CO 2 (g) + 2 H 2 O (g) Stoichiometry Anatomy of a Chemical Equation Reactants appear on the left side of the equation. CH 4 (g) + …
Bond Dissociation Energies of Organic Molecules
www2.chemistry.msu.eduBond Dissociation Energies of Organic Molecules STEPHEN J. BLANKSBY*,† AND G. BARNEY ELLISON*,‡ Department of Chemistry, University of Wollongong, NSW, 2522, Australia, and Department of Chemistry &
Chapter 1 Structure and Bonding - Chemistry
www2.chemistry.msu.eduThe atomic mass (atomic weight) of an element is weighted average mass in atomic mass units (amu) of an element’s naturally occurring isotopes. Carbon: Atomic Number and Atomic Mass 12C 6 AC Z 13C 6 (98.9 12.000) (1.1 13.000) 12.011 100 × +× =
Chapter 9: Atomic Absorption Spectrometry
www2.chemistry.msu.eduChapter 9: Atomic Absorption Spectrometry. Read: pp. 230 – 249 Problems: 9-1,3,5,6,8. Figure 9-13a. A(λ)= ε(λ)bC = log P. o /P
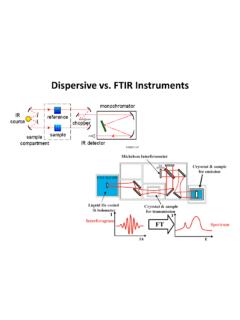
Dispersive vs. FTIR Instruments - Chemistry
www2.chemistry.msu.eduA spectroscopic technique used to observe vibrational, rotational, and other low-frequency [modes in a system. 1] It relies on inelastic scattering, or Raman scattering, of monochromatic light, usually from a laser in the visible, near infrared, or near ultraviolet range.
Principles of FTIR Spectroscopy - Chemistry
www2.chemistry.msu.eduPrinciples of Diffuse Reflectance Method K is the absorption coefficient, and S is the scattering coefficient. In practice, the comparative reflectance r∞ with respect to a standard powder such as KBr or KCl, of which K is near zero (0) in the actual measurement range Spectrum of solid caffeine Measurement of chemicals adhering to a surface ...
Chapter 5 Signals and Noise - Michigan State University
www2.chemistry.msu.eduNoise pick-up and possible amplification by the instrument circuit can be minimized. Difference Amplifiers →Analyte signal and reference signals are fed into the inputs of an operational amplifier is a scaler-type configuration. Common mode noise rejection. V o = R. k. R. i (V. 2 - V. 1)
Related documents
Chapter 17: Alcohols and Phenols - Vanderbilt University
as.vanderbilt.eduFigure 10.5 (Chapter 10.10) CC CC COH CO C O OR CO 2 CNH 2 C NH C C Cl l Cl CCl Cl Cl CCl Cl Cl Cl Increasing oxidation state 89 17.5: Alcohols from reduction of carbonyl compounds add the equivalent of H 2 across the π-bond of the carbonyl to give an alcohol RR' C O[H] R' RH H aldehyde (R or R´= H) → 1° alcohol ketone (R and R´≠ H ...
Chapter 4: Calculations Used in Analytical Chemistry
web.iyte.edu.trChapter 4: Calculations Used in Analytical Chemistry . 4A Some important units of measurement 4A-1 SI Units SI is the acronym for the French “Système International d’Unités.” The International System of Units (SI) is based on 7 fundamental base units.
Chapter 22 – Introduction to Electroanalytical Chemistry
www2.chemistry.msu.eduChapter 22 – Introduction to Electroanalytical Chemistry • Electroanalytical methods are a class of techniques in analytical chemistry, which study an analyte by measuring the potential (volts) and/or current (amperes) in an electrochemical cell containing the analyte.
Organic Chemistry II / CHEM 252 Chapter 20 – Amines
alpha.chem.umb.eduThe lone pair of the nitrogen atom accounts for most chemistry of amines – The unshared electron pair can act as a base or as a nucleophile • The nitrogen lone pair can also make a carbon nucleophilic by resonance. 30 ... Microsoft PowerPoint - Lecture_Chapter_20_Solomon
CHAPTER 12: COORDINATION CHEMISTRY IV: REACTIONS …
www.chem.uci.edu174 Chapter 12 Coordination Chemistry IV: Reactions and Mechanisms Copyright © 2014 Pearson Education, Inc. With two mechanistic pathways leading to the same product ...
Chapter 1 Organic Compounds: Alkanes
www.angelo.eduChapter 1 Alkanes 3 5 What is Organic Chemistry? • Organic chemistry is concerned with the study of the structure and properties of compounds containing carbon. – All organic compounds contain carbon atoms. – Inorganic compounds contain no carbons. Most inorganic compounds are ionic compounds. • Some carbon compounds are not considered ...
CHAPTER 6: ACID-BASE AND DONOR-ACCEPTOR CHEMISTRY
www.chem.uci.edu84 Chapter 6 Acid-Base and Donor-Acceptor Chemistry Copyright © 2014 Pearson Education, Inc. [HPPh3] relative to [HNPh 3]The conjugate acid [HPPh 3] has less steric ...
Organic Chemistry II / CHEM 252 Chapter 14 – Aromatic ...
alpha.chem.umb.eduOrganic Chemistry II / CHEM 252 Chapter 14 ... Nobel Prize in Chemistry 1996. Heterocyclic Aromatic Compounds 26 • Heterocyclic compounds have an element other than carbon in the ring • Example of aromatic heterocyclic compounds are shown below – …