Search results with tag "Chemistry"
Data Sheet for Chemistry A
ocr.org.ukGCE Chemistry A (Yellow) DC (SLM/SW) 15341/2 * OC E / 1 53 41* Data Sheet for Chemistry A (version 2.0) GCE Advanced Level and Advanced Subsidiary Chemistry A (H034, H434) Chemistry A units F321–F326 The information in this sheet is for the use of candidates following Chemistry A (H034 and H434).
The Basics of Water Chemistry (Part 1) - Pureflow
www.pureflowinc.comThe Basics of Water Chemistry (Part 1) By: C.F. “Chubb” Michaud Summary: Water chemistry is basic but, nonetheless, it’s still chemistry. Some people shy away from trying to understand his subject because they feel it’s over their heads. However, understanding the fundamentals
AP Chemistry Practice Exam from the 2018 Administration
m.shsbnu.netAP Chemistry Exam . 2017–18 AP Exam Instructions. Students are not allowed to use calculators in Section I of the AP Chemistry Exam. However, students are permitted to use scientiic or graphing calculators to answer questions in Section II. Four-function calculators are also permitted for use in Section II, but are not recommended.
Degrees in Chemistry - Royal Society of Chemistry
www.rsc.org* The economic benefits of higher education qualifications. Price Waterhouse Coopers LLP January 2005 3 Independent research * shows that the average chemistry graduate earns substantially more over a lifetime than graduates of many other disciplines: £190,000 more than those with two or more A-levels (no degree)
INTRODUCTION TO ATMOSPHERIC CHEMISTRY
projects.iq.harvard.eduIntroduction to Atmospheric Chemistry (Princeton University Press, 1999). They are arranged following the different chapters of the book. In recent years I have added course segments on aerosol chemistry and on mercury. I have included problems to support these course segments. All problems are from recent exams in my course. This 12 th
[CLASS XII CHEMISTRY PRACTICALS] - Saint John's Senior ...
stjohnsschoolkota.edu.in[CLASS XII CHEMISTRY PRACTICALS] Evaluation Scheme for Examination Marks Volumetric Analysis 08 Salt Analysis 08 Content Based Experiment 06 Project Work 04 Class record and viva 04 Total 30 Note:- 1. Chemical Equations of Experiment 3 to 11 are to be written on blank pages. ... CO -, ,-, , (CHEMISTRY PRACTICALS) NH ,
APPLICATIONS OF GREEN CHEMISTRY - IJSER
www.ijser.orgThis paper explain ideology, certain examples and application of green chemistry in everyday life, in industry, the laboratory and in education. Keywords: Clean chemistry, green catalyst, Environment, Eco-friendly Dry Cleaning, microwave preparations . 1. INTRODUCTION . Pharmaceuticals Industries are using toxic
A Level Chemistry Mark Scheme Paper 1 - Edexcel
qualifications.pearson.comMark Scheme (Results) Summer 2016 Pearson Edexcel GCE in Chemistry (8CH0) Paper 01 Core Inorganic and Physical Chemistry . Edexcel and BTEC Qualifications Edexcel and BTEC qualifications are awarded by Pearson, the UK’s largest awarding body. We provide a wide range of qualifications including academic,
Mark Scheme (Results) January 2013 - Edexcel
qualifications.pearson.comMar 07, 2013 · Chemistry (4CH0) Paper 2C Edexcel Level 1/Level 2 Certificate Chemistry (KCH0) Paper 2C . ... If you have any subject specific questions about this specification that require the help of a subject specialist, you can speak directly to the subject team at Pearson.
NC COMMUNITY COLLEGE COURSES NC A&T STATE …
www.ncat.eduANT 230 Physical Anthropology GEN ED: Social/Behavioral Science SOCI Elective Physical Anthropology ... BIO 120 Introductory Botany GEN ED: Natural Science BIOL 240 General Botany BIO 130 Introductory Zoology GEN ED: Natural Science BIOL 160 General Zoology ... BIO 180 Biological Chemistry Pre-Major/Elective BIOL Elective Biological Chemistry
ADVERTISEMENT 15
jobassam.in5 ECG Technician 6 2 0 1 0 2 UR-1 0 6 Electrician 10 3 1 1 1 2 UR-1 1 7 ICU Technician 47 19 3 5 2 12 UR-1, OBC-1 4 ... Pharmacy Council Pharmacy (Ayur) from Rs.60,500 Government recognized institute and must be ... Bio-Chemistry or Rs.60,500 Microbiology or Dairy Chemistry or Food Technology and Food & Nutrition recognized
BIOENGINEERING Catalog Year: 2021
student.engr.ucr.eduOverview of Bioengineering Organic Chemistry Organic Chemistry MATH 046 4 CS 009P (or CS 010A) 4 MATH 010B 4 Differential Equations Python (or C++ Programming) Multivariable Calculus ... Course Course Title (Units) ... BIEN 142 Introductory Biomedical Optical Imaging (4) BIEN/CEE 159 Dynamics of Biological Systems (4) BIEN 160 Biomedical ...
Organic Chemistry Practice Problems
www.tcc.fl.eduOrganic Chemistry Practice Problems (vii) Which substrate reacts faster via SN 2: (a) 1-bromo-1-methylcyclohexane (b) 1-bromo-2-methylcyclohexane 6) Provide an efficient multistep synthesis for each of the following conversions of the given starting material into product. For each transformation, give all necessary reagents and catalysts and give a
Volume Measurements and the Determination of Density
www.york.cuny.eduAn Introduction to Experimental Chemistry: Chemistry 101. York College of The City University of New York. 3 -York College Chem 101 Laboratory Fall 2009 PROCEDURE Determination of the Density of an Unknown Metal 1. Obtain an unknown metal sample and weigh it on the quadruple beam balance. Record
AP CHEMISTRY REVIEW PARTICULATE DIAGRAMS
intro.chem.okstate.eduAP CHEMISTRY REVIEW ... 2016 AP Q1 . Gelder/McGaw 3 2015 Q4 Show work: Name Section 35 1. Below are 1.0 L containers for the initial and equilibrium condition for the reaction, A 2 (g)! D 2 (g)! 2AD(g) Calculate the magnitude of the equilibrium constant for the reaction. Container Before Reaction Started
ap12 chemistry scoring guidelines - College Board
secure-media.collegeboard.orgAP® Chemistry 2012 Scoring Guidelines The College Board ... The partial pressures of these gases at 127 C are 0.600 atm for CO2(g) and 0.800 atm for H2O(g). ... On the basis of your answers above, write the balanced chemical equation for the combustion reaction and
Pearson Centre Number Candidate Number Edexcel GCE …
qualifications.pearson.comMay 23, 2018 · Chemistry Advanced Subsidiary Paper 1: Core Inorganic and Physical Chemistry Tuesday 22 May 2018 – Morning Time: 1 hour 30 minutes 8CH0/01 Candidates must have: Scientific calculator Data Booklet Pearson Edexcel GCE. 2 *P51459A0224* DO NOT RITE IN THIS AREA DO NOT RITE IN THIS AREA
SUMMER 2022 A LEVEL EXAMS TIMETABLE
www.bedales.org.uk13/06/2022 Pearson A Level 9CH0 01 Chemistry Paper 1: Advanced Inorganic and Physical Chemistry AM 1h 45m 13/06/2022 Pearson A Level 9EC0 03 Economics A Paper 3: Microeconomics and Macroeconomics PM 2h 13/06/2022 OCR A Level H443/03 Latin Prose literature PM 2h 14/06/2022 OCR A Level H573/02 Religious Studies Religion and ethics AM 2h
CLASS - XII CHEMISTRY Metallurgy - Yola
12-e.yolasite.comCLASS - XII CHEMISTRY (General principles and processes of isolation of elements) Topic :- Occurrence of metals, concentration methods Metallurgy: The scientific and technological process used for isolation of the metal from its ores is known as metallurgy.
NCERT Solutions for Class 11 Chemistry Chapter 11 - The p ...
cdn1.byjus.comNCERT Solutions for Class 11 Chemistry Chapter 11 The p-Block Elements Q 11.15 If B–Cl bond has a dipole moment, explain why BCl 3 molecule has zero dipole moment. Ans: The B – Cl bond is naturally polar because of the difference in the electronegativities of Cl and B.
Chemical Engineering Advising Handbook - che.psu.edu
www.che.psu.eduAug 31, 2021 · CHE 442 (MATSE 448) Polymer Processing Technology . CHE 443. Introduction to Polymer Science CHE 445 Bioremediation/Green Chemistry . CHE 444 Chemical Game Theory . CHE 445 Bioremediation/Green Chemistry . CHE 446. Transport Phenomena . CHE 449. Bioseparations . CHE 450. Process Dynamics and Control CHE 455 Drug Delivery, …
VCE Student Resources 2022 - VCE Exam Revision Lectures ...
www.tssm.com.auTYS-VS are powerful study, revision and consolidation tools that improve the students’ ... 3. Assessment & Exam Preparation (sample essay topics, final exam advice, glossary, references) Text Summary Notes for Text List 2 - Paired Comparisons ... 4 - Exam $8.00 Chemistry - Unit 1 - Exam 1 $8.00 Chemistry - Unit 2 - Exam 2 $8.00
ζ Î Q ¯ ÈÎ Ä ¶ÓÄÈ Q Ó©
tocc.eduChemistry. Hamidou Keita MW 03:00 PM 04:30 PM 05/23/2022 07/18/2022 Virtual via ZOL ONLIN OLINS oom CHM 121N 1 4.00 Chemistry and Society CHM 121N 1. ... Daniel Raymond Sestiaga, Jr. 00:00 AM 00:00 AM 05/23/2022 07/18/2022 TBD CPH 101 1 2.00 Exploring Careers in Health CPH 101 1.
Blood Chemistry & CBC Analysis
www.functionalmedicineuniversity.compatient‟s history and physical exam to form a working diagnosis. It may also be used in decision making for advanced diagnostic testing. Note: It is extremely important that you are registered with CLIA (Clinical Laboratory Improvement Amendments) before you begin to perform in-office testing. A 1998 Amendment expanded earlier regulations to
AP Chemistry 2010 Free-Response Questions - College Board
secure-media.collegeboard.org2010 Free-Response Questions The College Board The College Board is a not-for-profit membership association whose mission is to connect students to college success and opportunity. Founded in 1900, the College Board is composed of more than 5,700 schools, colleges, universities and other educational organizations.
AP Chemistry 2011 Scoring Guidelines - College Board
secure-media.collegeboard.org2011 Scoring Guidelines . The College Board . The College Board is a not-for-profit membership association whose mission is to connect students to college success and opportunity. Founded in 1900, the College Board is composed of more than 5,700 schools, colleges, universities and other educational organizations.
GCE Chemistry A
ocr.org.ukH432/03 Mark Scheme June 2018 11 Question Answer Marks Guidance 3 (a) Overall 3– charge shown (outside brackets) for at least ONE isomer 3– must apply to the overall charge of structures F e O O O O O O 3± O O O O O O F e O O O O O O 3± O O O O O O For bond into paper, 1 mark for each isomer Bonds must go to O ligand atoms on EACH structure
3 PERIODIC TABLE AND PERIODICITY IN PROPERTIES
www.nios.ac.inNotes 77 Periodic Table and Periodicity in Properties CHEMISTRY MODULE - 2 Atomic Structure and z co-relate the sequence of arrangements of elements in periodic table with Chemical Bonding electronic configuration of the elements; z recall the designations of the groups (1-18) in the periodic table; z locate the classifiction of elements into s-, p-, d- and f- blocks of the periodic
Platinum Business Studies Navigation Pack Grade 10 - Pearson
za.pearson.comAt Pearson South Africa, we believe that education is the key to every individual’s success. ... Paper 2 Chemistry Page 56 TOTAL HOURS = 25 *10 This topic has been moved from term 1 to term 4. This topic is on pages 84–90 ... All Grade 9 topics Baseline assessment to determine learning losses in Grade 9 3 Hours Completed in week 1 of Term 1
8 The Variational Principle - School of Physics and Astronomy
www2.ph.ed.ac.ukIn practice, this is how most quantum mechanics problems are solved. ... the binding energy of a deuteron due to the strong nuclear force, with A=32MeV and a=2.2fm. The strong nuclear force does not exactly have the form V(r) ... chemistry, materials, minerals and beyond.
2000 AP Chemistry Scoring Guidelines - College Board
secure-media.collegeboard.org2000 Scoring Guidelines These materials were produced by Educational Testing Service (ETS), which develops and administers the examinations of the Advanced Placement Program for the College Board. The College Board and Educational Testing Service (ETS) are dedicated to the principle of equal opportunity, and their programs, services, and
Peterson’s Test Prep Spring 2022 Test Prep: Advanced ...
terc-cdn.petersons.comPractice Tests AP Chemistry Practice Tests 2 R 2022 2020 Practice Tests AP Computer Science Practice Tests 3 R 2022 2020 ... Practice Tests Postal Worker 710/711 1 £ 2016 N/A Practice Tests Praxis Core Practice Tests 1 ...
CHAPTER5 Periodic Classification of Elements
ncert.nic.inPeriodic Table. The Periodic Table proved to be the unifying principle in chemistry. It was the motivation for the discovery of some new elements. When Mendeléev started his work, 63 elements were known. He examined the relationship between the atomic masses of the elements and their physical and chemical properties. Among chemical properties,
bk technologies kng accessory catalog
www.bktechnologies.comSingle Bay Smart Charger. Multi-chemistry capable: NiCad, NiMH, Li-Ion and Li-Po Adaptive reconditioning capability. Display Showing Battery’s Charge or Life Condition Wide Range Power Input: AC Input (110 –240V AC) or DC Input (14 – 24v …
2004 AP Chemistry Free Response Questions - College Board
secure-media.collegeboard.org21 39 88.91 57 138.91 89 227.03 22 40 91.22 72 178.49 23 41 92.91 73 180.95 24 42 95.94 74 183.85 25 43 (98) 75 186.21 26 44 101.1 76 190.2 27 45 102.91 77 192.2 28 46 106.42 78 195.08 29 47 107.87 79 196.97 30 48 112.41 80 200.59 5 13 31 49 114.82 81 204.38 6 14 32 50 118.71 82 207.2 7 15 33 51 121.75 83 208.98 8 16 34 52 127.60 84 (209) 9 17 ...
Mark Scheme (Results) January 2021 - IG Exams
igexams.comPearson Edexcel International GCSE . In Chemistry (4CH1) Paper 1C and Science (Double Award) (4SD0) Paper 1C www.igexams.com. Edexcel and BTEC Qualifications . Edexcel and BTEC qualifications are awarded by Pearson, the UK’s largest awarding body. We provide a wide range of qualifications including academic, vocational, occupational and ...
Introduction to Polymer Science (Book) - Chemistry Is All ...
chemistry.pixel-online.orgINTRODUCTION TO POLYMER SCIENCE 1 Polymer science was born in the great industrial laboratories of the world of the need to make and understand new kinds of plastics, rubber, adhesives, fibers, and coatings. Only much later did polymer science come to academic life. Perhaps because of its origins, polymer science tends to be more inter-
SAMFORD UNIVERSITY 2021 ANNUAL REPORT
www.samford.eduthe university’s Advanced Placement STEM Success project. The program seeks to increase the self-efficacy of students from historically marginalized backgrounds by providing elevated educational opportunities and support for success in AP biology and chemistry course work. Samford University School of the Arts and Miles . College launched the
The Ministry of Health, Labour and Welfare Ministerial ...
www.mhlw.go.jpG1 Physics and Chemistry Validation of Analytical Procedures <G1-1-130>.....2607 System Suitability <G1-2-152> .....2609 Near Infrared ... Radioactivity Measurements Method for Crude Drugs <G5-8-180>.....2731 G6 Drug Formulation Criteria for Content Uniformity in Real Time ...
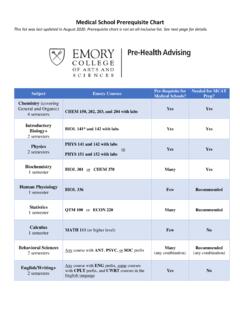
Medical School Prerequisite Chart - Emory University
prehealth.emory.edu• You have questions about using your AP/IB credit • You began Chemistry coursework at Emory prior to Fall 2017 and have questions about how to proceed • You transferred to Emory and have questions about prerequisite coursework taken at your previous institution *If you use AP credit for BIOL 141, you must still take the BIOL 141 Lab.
11th & 12th Grade Elective Course Selection Sheet
www.esasd.netMathematics (.05 Credit) Chemistry (CP, Honors & AP) 467 SAT Prep Math Biology II (Applied, CP, Honors ) 468 SAT Prep Math - Honors Anatomy & Physiology (Applied, CP, Honors) Gifted IEP Physics (CP, Honors & AP) Enrichment Program - (1 credit) Environmental Science (Applied & CP) 892 Honors Seminar Please schedule a conference with your Biology ...
Course modifications 2021–22: Advanced Higher Chemistry
www.sqa.org.ukRevision support for exam: Study guide A summary table that shows the modifications we made to each course, alongside the revision support, is available on SQA’s website. We published more detailed modification documents for schools, colleges and training providers in August 2021. If you have any questions about the modifications for this course
Chemistry Investigatory Project - Weebly
neerajminichemistry.weebly.comThis is to certify that this “Chemistry Investigatory Project” has been successfully completed by Dhananjay Dhiman of class XII – A under the guidance of Mr. B. Venkaiah in particular fulfilment of the curriculum of Central Board of Secondary Education {CBSE} leading to the award of annual examination of the year 2016-17.
Chemistry - Edexcel
qualifications.pearson.comMay 25, 2016 · Paper 2: Core Organic and Physical Chemistry Friday 10 June 2016 – Afternoon Time: 1 hour 30 minutes 8CH0/02 You must have: Data Booklet Scientific calculator, ruler Instructions •• Use black ink or ball-point pen. Fill in the boxes at the top of this page with your name, centre number and candidate number.• • Answer all questions.
Chemistry: The Periodic Table and Periodicity
mrsklattscience.weebly.comNOBLE GASES 23. What is the name given to the group of elements that have the following valence shell electron configurations? a. s2 b. s2p6 c. s2p5 d. s1 ALKALINE EARTH METALS NOBLE GASES HALOGENS ALKALI METALS 24. List the three lightest members of the noble gases. He, Ne, Ar 25. List all of the alkali metals. Li, Na, K, Rb, Cs, Fr 26.
Chemistry 51 ASNWER KEY REVIEW QUESTIONS
mymission.lamission.eduREVIEW QUESTIONS Chapter 6 1. Classify the type of each of the following reactions: a) (NH 4) 2 SO 4 fi 2 NH 3 + SO 2 + H 2 decomposition b) Br 2 + 2 KI fi 2 KBr + I 2 single replacement c) 2 Na + Cl 2 fi 2 NaCl synthesis d) Al(OH) 3 + 3 HCl fi AlCl 3 + 3 H 2 O double replacement 2.
Similar queries
Chemistry, The Basics of Water Chemistry Part, AP Chemistry, 2017, Royal Society of Chemistry, Coopers, INTRODUCTION, CLASS XII CHEMISTRY PRACTICALS, CLASS, APPLICATIONS OF GREEN CHEMISTRY, A Level Chemistry Mark Scheme, Mark Scheme, Specification, COMMUNITY COLLEGE, Physical, Introductory, Technician, Pharmacy, Organic Chemistry Organic Chemistry, Course Course Title, Organic Chemistry, Chemistry 101, City University of New York, PARTICULATE DIAGRAMS, 2016 AP, Gases, Answers, Physical Chemistry, Level, Chemistry Paper, Paper, Solutions for Class 11 Chemistry Chapter 11, Exam, Study, Final exam, Raymond, Blood Chemistry & CBC Analysis, Clinical, Chemistry 2010, 2010, Guidelines, Universities, Mark, Notes, Grade, Pearson, Variational Principle, Practice, Problems, Nuclear, 2016, Periodic, Elements, Periodic Table, Charger, Multi, Battery, Edexcel International, Edexcel, Industrial, Advanced Placement, Radioactivity, Emory, Honors, Biology, Physics, Study guide, Chemistry Investigatory Project, Periodicity, Chemistry 51 ASNWER KEY REVIEW QUESTIONS, REVIEW QUESTIONS





![[CLASS XII CHEMISTRY PRACTICALS] - Saint John's Senior ...](/cache/preview/8/e/c/c/7/0/2/b/thumb-8ecc702b441a6099d9810dbf9f2c3c2f.jpg)