Tutorial 3 THE MOLE AND STOICHIOMETRY
Tutorial 3 THE MOLE AND STOICHIOMETRY A chemical equation shows the reactants (left side) and products (right side) in a chemical reaction. A balanced equation shows, in terms of moles, how much of each substance is involved in the reaction. Stoichiometry is the study of the relationships of quantities of substances in a chemical reaction.
Download Tutorial 3 THE MOLE AND STOICHIOMETRY
Information
Domain:
Source:
Link to this page:
Please notify us if you found a problem with this document:
Documents from same domain
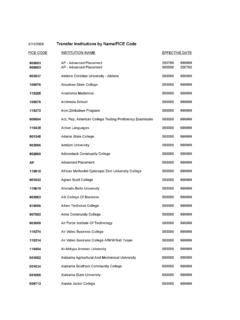
Transfer Institutions by Name/FICE Code - EIU
www.eiu.eduINSTITUTION INAME EFFECTIVE DATE 5/15/2008 FICE CODE Transfer Institutions by Name/FICE Code 001061 Alaska Pacific University 000000 999999 002885 Albany College Pharmacy 000000 999999
Name, Code, College, Institutions, Transfer, Fice, Transfer institutions by name fice code
Designing and Conducting Focus Group Interviews …
www.eiu.eduFocus Group Interviewing --- Richard Krueger 4 Beginning the Focus Group Discussion The first few moments in focus group discussion are critical.
Group, Discussion, Focus, Focus group, Focus group discussion
Acceptance Letter Sample - Eastern Illinois University
www.eiu.eduAcceptance Letter Sample 1435 Lincoln Ave Charleston IL 61920 Today’s Date Ms. Mary Lou Nelson Manager of Human Resources XYZ Corporation
Illinois, University, Samples, Letter, Acceptance, Easterns, Acceptance letter sample eastern illinois university, Acceptance letter sample
Business Administration Minor - EIU
www.eiu.eduBusiness Administration Minor is a great supplement to your major, ... FIN 3770 Working Capital Management. Non-business majors--including students enrolled in the
Business, Administration, Management, Minor, Business administration minor
Best Practices in Academic Assessment - EIU
www.eiu.eduAssessment is most likely to lead to improvement when it is part of a larger set of conditions that promote change. Assessment alone changes little.
Texas Bandits: A Study of the 1948 Democratic Primary
www.eiu.edu1948, Lyndon Johnson unleashed an aggressive, modern, and crooked campaign because his political career stood in the balance. In 1941, Johnson led by 5,000 votes through a count of 96% of the ballots.
Study, Primary, Democratic, 1948, Study of the 1948 democratic primary
Timeline of Notable Scientists in Physics and Chemistry
www.eiu.eduTimeline of Notable Scientists in Physics and Chemistry 1 Timeline of Notable Scientists in Physics and Chemistry . Overview: To review, students will match the names of scientists with their respective contributions to science. The teacher will guide the students through the correct chronological sequence of achievements.
Scientist, Physics, Timeline, Notable, Timeline of notable scientists in physics and
www.eiu.edu
www.eiu.eduCreated Date: 4/21/2014 9:20:12 AM
Illinois workNet® Quick Start Guide - EIU
www.eiu.eduIllinois workNet® Quick Start Guide An online system, a mobile app, and a network of locations throughout your community helping individuals, employers, and
Guide, Network, Illinois, Quick, Start, 174 quick start guide, Illinois worknet
What does the Epic of Gilgamesh Reveal about …
www.eiu.edusociety of Mesopotamia nearly 4,000 years ago. Gilgamesh is known to be the first great hero, and the epic is known as the ‘first great masterpiece of world literature’.
About, Reveals, Mesopotamia, Gilgamesh, Of gilgamesh reveal about
Related documents
Gas Stoichiometry Chemistry 110 - Cerritos College
www.cerritos.eduGas Stoichiometry . Chemistry 110. 1] Given the equation: 2 NH3 (g) + 3 Cl 2(g) ---> N (g) + 6 HCl(g) a. How many milliliters of nitrogen can be made from 13 L of chlorine and 10.0 L of ammonia gas at STP? 10.0 L NH3 X . 1 L N. 2. 2 L NH3 = 5.00 L N. 2.
Chapter 13 Stoichiometry - Glendale Community College
web.gccaz.eduClark, Smith (CC-BY-4.0) GCC CHM 130 Chapter 13: Stoichiometry page 3 13.4 Volume-Volume Stoichiometry Molar Volume gas @ STP Fact: If you start with liters of the given and are asked to find liters of the unknown, as long as the gases are at the same temperature and pressure the molar volumes will cancel out with each other so you are ...
Moles And Stoichiometry Practice Problems Answers - MS.
msswartzscience.weebly.comAnswers: Moles and Stoichiometry Practice Problems 1) How many moles of sodium atoms correspond to 1.56x1021 atoms of sodium? 1.56 -x 1021 atoms Na x 1 mol Na = 2.59 x 10 3 mol Na 236.022 x 10 atoms Na 2) Determine the mass in grams of each of the following: a. 1.35 mol of Fe 1.35 mol Fe x 55.845 g Fe = 75.4 g Fe
STOICHIOMETRY AND PERCENT PURITY Many samples of …
www.kpu.caSTOICHIOMETRY AND PERCENT PURITY Many samples of chemicals are not pure. We can define percent purity as mass of pure compound in the impure sample total mass of impure sample ... 2 needed to make 127 g of Mg3(PO4)2, assuming a 100% yield. mass Mg(OH)2 = 127 g Mg3(PO4)2 x 1 mole Mg3(PO4)2 x 58.3 g Mg(OH)2 1 mole Mg(OH)2 1 mole Mg3(PO4) 3 …
Many, Percent, Purity, Stoichiometry, Stoichiometry and percent purity many
Equation Stoichiometry Chemistry 110 - Cerritos College
www.cerritos.eduEquation Stoichiometry . Chemistry 110 . 1] Given the equation: 2C8H18 + 25 O2----> 16CO2 + 18H2O . a. How many moles of oxygen gas are required to make 8.33 moles of carbon dioxide? ... How many moles of Al4C3 were reacted when 3.55 x 10 35 formulas units of aluminum hydroxide were produced? 3.55 x 1035 formula units Al(OH)3 X . 1mol Al(OH)3 6 ...
Limiting Reagent Worksheet - Everett Community College
www.everettcc.edu3 PO 4 x 163.94 g Na 3 PO 4 = 126.75 g FeCl 2 3 mole FeCl 2 1 mole Na 3 PO 4 = 20. g Na 3 PO 4 Since we have 41 g Na 3 PO 4, FeCl 2 is the limiting reagent. 23 g FeCl 2 x 1 mole FeCl 2 x 6 mole NaCl x 58.44 g NaCl = 126.75 g FeCl 2 3 mole FeCl 2 1 mole NaCl = 21 g NaCl 3) How much of the excess reagent remains when this reaction has gone to ...
Worksheet, Community, College, Grantee, Everett, Everett community college, Limiting, Limiting reagent worksheet
Stoichiometry - Montgomery College
www.montgomerycollege.eduReaction Stoichiometry Moles of The coefficients in a balanced chemical equation specify the relative amounts in moles of each of the substances involved in the reaction. 2 C8H18 (l) + 25 O2(g) 16 CO2 (g) + 18 H2O(g) 2 mol C8H18: 25 mol O2: 16 mol CO2: 18 mol H2O How many grams of glucose can be synthesized from 37.8 g of CO2 in photosynthesis?