Dihydrogen Monoxide
Found 9 free book(s)Naming Covalent Compounds Worksheet Key
www.humbleisd.netCarbon monoxide Dihydrogen monoxide Dihydrogen dioxide Nitrogen trihydride Dicarbon dihydride Dinitrogen tetrahydride Sulfur trioxide Dichlorine monoxide Carbon tetrahydride Dinitrogen monoxide Sulfur dioxide . 24. Tetracarbon decahydride 25. Chlorine trifluoride PART Il: Write the names for the followin covalent compounds: 10. 11. 12. 13.
Common Cations, Anions, Acids, Salts and Hydrate ...
web.mst.edu3 Ozone NO Nitrogen monoxide (Nitric Oxide) 4 Tetra H 2O Water (Dihydrogen Monoxide) NO 2 Nitrogen dioxide 5 Penta F 2 Fluorine N 2O Dinitrogen monoxide (Nitrous oxide) 6 Hexa HF Hydrogen fluoride N 2O 2 Dinitrogen dioxide 7 Hepta Cl 2 Chlorine N 2O 4 Dinitrogen tetroxide 8 Octa HCl Hydrogen chloride CO Carbon monoxide 9 Nona Br 2 Bromine CO 2 ...
Naming Ionic Compounds – Answer Key - Weebly
siemianowski.weebly.com12. CO carbon monoxide 13. H2O dihydrogen monoxide, water 14. FeO iron (II) oxide 15. NaCl sodium chloride 16. SO3 sulfur trioxide 17. BaO barium oxide 18. NH3 nitrogen trihydride, ammonia 19. CO2 carbon dioxide 20. NO nitrogen monoxide
Common Polyatomic Ions - West Virginia University
pltl.wvu.edugallium oxide Dihydrogen monoxide Provide the correct name for the following acids or bases. NaOH NH 3 H2SO 3 HCN H2S Ca(OH) 2 H3PO 4 Fe(OH) 3 Write the correct chemical formula for the following acids or bases. hydrofluoric acid cobalt (II) hydroxide hydroselenic acid sulfuric acid chlorous acid beryllium hydroxide
HYDROGEN - NCERT
ncert.nic.inThe production of dihydrogen can be increased by reacting carbon monoxide of syngas mixtures with steam in the presence of iron chromate as catalyst. ( ) ( ) 673K ( ) ( ) COgHOg COgHg+⎯⎯⎯⎯ 222catalyst →+ This is called water-gas shift reaction. Carbon dioxide is removed by scrubbing with sodium arsenite solution. Presently ~77% of the ...
HYDROGEN - NCERT
www.ncert.nic.inThe production of dihydrogen can be increased by reacting carbon monoxide of syngas mixtures with steam in the presence of iron chromate as catalyst. ( ) ( ) 673K ( ) ( ) COg HOg+ → +2 catalyst OgC Hg22 This is called water-gas shift reaction. Carbon dioxide is removed by scrubbing with sodium arsenite solution.
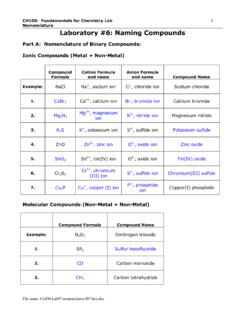
Laboratory #6: Naming Compounds
spot.pcc.edudihydrogen phosphate 5. Ag 3PO 4 silver phosphate 30. CoS cobalt(II) sulfide 6. H2S hydrosulfuric acid 31. Cd 3(AsO 4)2 cadmium arsenate (or hydrogen sulfide) 7. CaO calcium oxide 32. NaHSO 3 sodium hydrogen sulfite 8. H2CO 3 carbonic acid 33. Li2HPO 4 lithium hydrogen phosphate 9. Ni(NO 3)2 nickel(II) nitrate 34. H 3PO 3 phosphorous acid 10 ...
COVALENT
chemunlimited.comdinitrogen monoxide: N 2 O phosphorus pentachloride: PC1 5 sulfur hexafluoride: SF 6 diphosphorus pentoxide: P 2 O 5 Most organic compounds that contain alcohol (R-OH) or carboxylic acid (R-COOH) functional groups will most likely be soluble due to the hydrogen bonding capabilities of those substances. Other covalent compounds tend to be ...
WORKSHEET: Chemical Bonding – Ionic & Covalent!
www.johnbowne.orgDec 18, 2015 · PART 2: Use Lewis dot structures to show the ionic bonding in the following pairs of elements.Show the transfer of electrons using arrows. Write the correct chemical formula for the ionic compound that forms. 1) barium oxide (Ba and O) 4) sodium oxide (Na and O)