Search results with tag "Ceftriaxone"
Prescription Drug Guide October 2021
www.myprime.comceftriaxone sodium for inj 250 mg. 1: ceftriaxone sodium for inj 500 mg (Rocephin) 1. ceftriaxone sodium for inj 1 gm (Rocephin) 1. ceftriaxone sodium for inj 2 gm. 1: cefuroxime axetil tab 250 mg (Ceftin) 1. cefuroxime axetil tab 500 mg …
ANMC Pediatric Acute Otitis Media (AOM) Treatment …
anmc.orgCeftriaxone 50mg/kg IM or IV daily for 1-3 days (max 2000mg/dose) Ceftriaxone 50mg/kg IM or IV daily for 3 days (max 2000mg/dose) or 10mg/kg PO TID (max 450mg/dose) or PLUS (cefuroxime£, cefprozil, cefdinir or ceftriaxone) Supportive Medications Acetaminophen 15mg/kg PO q4-6hr PRN pain or fever, not to exceed 75mg/kg in 24 hours (max 4g in 24 ...
Ceftron - Square Pharmaceuticals
www.squarepharma.com.bdRegistered Trade Mark Ceftriaxone Ceftron® COMPOSITION Ceftron® 250 mg IM injection: Each 250 mg vial con tains dry substance equivalent to 250 mg Ceftriaxone (as sterile Ceftriaxone Sodium USP) accompanied by a solvent ampoule of 2 ml
Double Anaerobic Coverage: What is the role in clinical ...
www.unmc.educeftriaxone, ciprofloxacin PLUS Metronidazole Cefepime, ceftazidime, ciprofloxacin PLUS Metronidazole Ceftriaxone, cefotaxime, cefepime, ceftazidime PLUS ... historical perspective and review of the most recent data (2005-2007). Clin Infect Dis. 2010; 50 Suppl 1: S26-33 2.
Cetriaxone 1g 2g Powder for Soln for Inj PL 14894 0342-3 ...
www.mhra.gov.ukUKPAR Ceftriaxone Sodium 1g & 2g Powder for Solution for Injection PL 14894/0342-3 1 CEFTRIAXONE SODIUM 1G POWDER FOR SOLUTION FOR INJECTION
A RANDOMIZED, DOUBLE-BLIND, PLACEBO …
www.aldf.comclinical protocol phase iii a randomized, double-blind, placebo-controlled, multicenter trial of the safety and efficacy of ceftriaxone and doxycycline in thetreatment of patients with
PARENTERAL NUTRITION (PN) AND DRUG COMPATIBILITY …
www.pedmed.orgamphotericin ceftriaxone furosemide sodium bicarbonate ampicillin cisplatin mannitol ATG deferoxamineparaldehyde For any drugs not listed here, the prescribing physician should discuss with the pharmacist the information available on the drug and the risks/benefits of co-
薬剤耐性ワンヘルス動向調査年次報告書 2020
www.mhlw.go.jpAMED Japan Agency for Medical Research and Development ... CRE Carbapenem-resistant Enterobacteriaceae ... ceftriaxone CTRX cefoperazone/sulbactam CPZ/SBT cefdinir CFDN cefcapene pivoxil CFPN-PI cefditoren pivoxil CDTR-PI cefixime CFIX 第4世代
Children’s Health Queensland Paediatric Antibiocard ...
www.childrens.health.qld.gov.aureview). Immediate type hypersensitivity, Meropenem IV 40 mg/kg/dose IV every 8 hourly (maximum 2 g/dose) and seek ID . Febrile non-neutropenia (Oncology) Over 1 month of age: Ceftriaxone IV 100 mg/kg once daily (maximum 4 g/day) and discuss with Paediatric Oncologist. Refer to CHQ-GDL-01249 Management of Fever in a Paediatric Oncology
Multi-drug Resistant Organisms (MDROs) in Healthcare ...
www.ndhealth.govCeftriaxone R Colistin MIC >4 ... Antibiotic Review F441: Because of increases in MDROs, review of the use of antibiotics is a vital aspect of the infection prevention and control program. An area of increased surveyor focus- an area where you need to assess if you are meeting
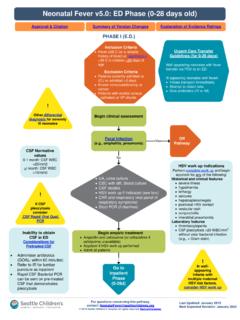
Neonatal Fever v8.0: ED Phase (0-28 days old)
www.seattlechildrens.orgNext Expected Review: January 2024 ... Ceftriaxone Admit to inpatient on UTI and Bronchiolitis pathway inpatient on UTI and Bronchiolitis CONSIDER BRONCHIOLITIS PATHWAY Signs of poor perfusion or mental status changes or sepsis score > 3 Continue workup per Neonatal
ANTIBIOTIC CROSS-SENSITIVITY CHART July 2018
www.vhpharmsci.comVA Pharmaceutical Sciences CSU PARENTERAL ANTIBIOTIC ALLERGY CROSS-SENSITIVITY CHART Amikacin Ampicillin Azithromycin Cefazolin Cefotaxime Cefoxitin Ceftazidime Ceftriaxone Cefuroxime Chloramphenicol Ciprofloxacin Clindamycin Cloxacillin Cotrimoxazole (Sulfa) Daptomycin Ertapenem Erythromycin Gentamicin Imipenem Levofloxacin Meropenem Metronidazole Moxifloxacin Penicillin …
Lesson 2: Understanding expressions of drug amounts
courses.washington.edusodium bicarbonate bismuth subcarbonate dissolve in 8 ounces of water and drink prn indigestion ... For example, ceftriaxone, an injectable antibiotic, is stated to have a solubility in water of 400mg/ml at 25°C (room temperature). This means that if you try to place 500mg in 1ml at 25°C, you will get a layer of stuff at the bottom of the vial.
CEFTRIAXONE-AFT - Medsafe
www.medsafe.govt.nzCEFTRIAXONE-AFT Ceftriaxone sodium equivalent to ceftriaxone 500 mg, 1 g and 2 g powder for injection Presentation Ceftriaxone-AFT is a white to pale yellow powder packed in vials which contain the
Ceftriaxone 2g IV infusion administration
www.wsh.nhs.ukReview date:14/7/24 Page No: 1 Outpatient Parenteral Antimicrobial Therapy (OPAT) Self-administration of Ceftriaxone 2g IV infusion This leaflet is designed to support patients, and nursing staff who are teaching patients to self-administer Ceftriaxone, with the assistance of the OPAT team. Please use this
Ceftriaxone – safety in neonates
www.who.intReview Ceftriaxone is used mostly in hospital practice for neonatal sepsis, meningitis and ophthalmia neonatorum. Its broad spectrum of activity, once daily dosing and good
Ceftriaxone – safety in neonates - WHO
www.who.intterm neonates with Gram negative bacterial meningitis treated with a third generation cephalosporin and amikacin [4]. In a study comparing cefotaxime and ceftriaxone for severe bacterial infections in children, 22 neonates were enrolled. Both were effective and adverse reactions were rare[5]. Other early studies also have shown it to be
Ceftriaxone - Antimicrobe
www.antimicrobe.orgCeftriaxone Antibiotic Class: Third-Generation Cephalosporin Antimicrobial Spectrum: Staphylococcus aureus (methicillin susceptible), Coagulase negative Staphylococci ...
Ceftriaxone- Resistant Neisseria gonorrhoeae, Japan
wwwnc.cdc.govLETTERS Ceftriaxone-Resistant Neisseria gonorrhoeae, Japan To the Editor: Spread of multi- drug-resistant Neisseria gonorrhoeae is a major public health concern. Ef-fective antimicrobial therapy is a key
Similar queries
Ceftriaxone sodium, ANMC Pediatric Acute Otitis Media (AOM) Treatment, Ceftriaxone, Ceftron, Ceftriaxone Ceftron, Double Anaerobic Coverage: What is, Review, Cetriaxone 1g 2g Powder for, DOUBLE, BLIND, PLACEBO, Clinical, Double-blind, placebo-controlled, multicenter, Ceftriaxone and doxycycline in thetreatment, Sodium, Japan, Resistant, Multi-drug Resistant Organisms (MDROs) in Healthcare, Neonatal Fever, ANTIBIOTIC CROSS-SENSITIVITY, Ceftriaxone-AFT, Ceftriaxone – safety in neonates, Neonates, Children, Ceftriaxone- Resistant Neisseria gonorrhoeae, Japan, Ceftriaxone-Resistant Neisseria gonorrhoeae, Japan