Search results with tag "Data integrity"
MHRA GMP Data Integrity Definitions and Guidance for ...
assets.publishing.service.gov.ukMHRA GMP Data Integrity Definitions and Guidance for Industry March 2015 . Introduction: Data integrity is fundamental in a pharmaceutical quality system which ensures that medicines are of the required quality. This document provides MHRA guidance on GMP data integrity expectations for the pharmaceutical industry. This guidance is intended to ...
و ةيساسالأ ئدابلما - SDAIA
sdaia.gov.saData Confidentiality: The state of keeping data secret by preserving authorized restrictions on data access and disclosure. Data Integrity: The state of ensuring data validity by guarding against improper information modification or destruction. Restricted / Protected Data: Data classified as Confidential, Secret, or Top Secret.
The ServiceNow Configuration Management Database
www.servicenow.comstores a complete history of discovered data from each source, you can easily switch sources at any time. Ensure data consistency and integrity ITOM Discovery works hand-in-handwith the ServiceNow CMDB to ensure data integrity and consistency. For example, it ensures that discovered data is mapped to the right location in the CMDB data model.
Medicines & Healthcare products Regulatory Agency (MHRA ...
assets.publishing.service.gov.ukdata integrity failures within the organisation’s systems. 3.6 The effort and resource applied to assure the integrity of the data should be commensurate with the risk and impact of a data integrity failure to the patient or environment. Collectively these arrangements fulfil the concept of data governance.
MHRA GMP Data Integrity Definitions and Guidance for ...
www.it-asso.comMHRA GMP Data Integrity Definitions and Guidance for Industry March 2015 . Introduction: Data integrity is fundamental in a pharmaceutical quality system which ensures that medicines are of
Practical risk-based guide for managing data integrity
apic.cefic.orgAchieving data integrity compliance, for paper, electronic and hybrid systems, requires translation of these principles into practical controls in order to assure GxP-impacting business decisions can be verified and inspected throughout the data lifecycle. Currently available regulatory guidelines have been used to elaborate the approach ...
GOOD PRACTICES FOR DATA MANAGEMENT AND …
picscheme.org2.4 Data Integrity is defined as “the extent to which all data are complete, consistent and ... required for data verification and evidence of operational compliance with 169 procedures. 170 4.3 Whilst this document has been written with the above scope, many principles
MHRA GMP Data Integrity Definitions and …
www.it-asso.comData integrity requirements apply equally to manual (paper) and electronic data. Manufacturers and analytical laboratories should be aware that reverting from automated / computerised to manual /
13 INFORMATION MANAGEMENT AND TECHNOLOGY
www.jblearning.comintegrity must be maintained so that current data are available when needed. If data integrity is less than optimal, it could result in inappropriate decisions that could possibly harm a patient.
Introduction to ISO Identification of Medicinal Products ...
www.ema.europa.euAdverse event reports are based on a harmonised set of product definitions, improving the quality of data used for signal management, and speeding up communication, ... Good Manufacturing Practices (GMP) inspections: ... Improving data integrity and reliability;
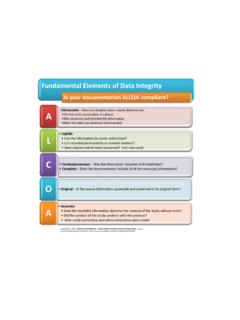
Fundamental Elements of Data Integrity
www.feinsteininstitute.orgTitle: Microsoft PowerPoint - Revised ALCOA (Electronic).pptx Author: ekim Created Date: 1/25/2011 12:25:21 PM
2017 GMP事例研究会 - jpma.or.jp
www.jpma.or.jp2017年度GMP事例研究会. BMS愛知工場における Data Integrityの取り組み事例 BMS K.K. 愛知工場 クオリティオペレーション 品質保証 杉浦 昇 1
Merck Corporate University - jpma.or.jp
www.jpma.or.jp•米国FDAやEU規制当局などは、データの完全性( Data Integrity : DI )に焦点を置いた監視を強化し、DI 強化のためのガイダンスを
DATA GOVERNANCE POLICY
unlv.eduData integrity refers to the validity, reliability, and accuracy of data. Data integrity relies on a clear understanding of the business processes underlying the data and the consistent defmition of each data element. Data integration, or the ability of …
Data Domain Invulnerability Architecture: Enhancing Data ...
www.emc.com3 | Data Domain Data Invulnerability Architecture © 2017 Dell Inc. or its subsidiaries. EXECUTIVE SUMMARY STORAGE SYSTEM DATA INTEGRITY Behind all their added value ...
Data Integrity Article - Ofni Systems
www.ofnisystems.comReview of Good Data Integrity Principles Page 6 of 11 WHO Guidance on Good Data and Record Management Practices The World Health Organization (WHO) is an agency of the United Nations concerned with international
Data Integrity and Good Documentation Practices: …
asq.orgDefinitions October 12 - 13, 2017 26th Annual ASQ Audit Division Conference: The Intercontinental Addison Data Integrity means the extent to which all data are complete, ...
Similar queries
GMP Data Integrity Definitions and Guidance for, GMP Data Integrity Definitions and Guidance for Industry, Data Integrity, Guidance, GMP data integrity, Industry, Data, History, Integrity, Data Integrity Definitions and Guidance for, Data integrity compliance, Compliance, Data Integrity Definitions and, 13 INFORMATION MANAGEMENT AND TECHNOLOGY, Current data, Definitions, Good Manufacturing Practices, Data Domain Data Invulnerability Architecture, Data Integrity Article, Conference