Search results with tag "Amiodarone"
Suivi de la TSH et amiodarone - Réalités …
www.realites-cardiologiques.comSuivi de la TSH et amiodarone SUIVI DE LA TSH SOUS AMIODARONE La figure 1 résume la surveillance d’un patient sous amioda- rone. Le dosage de TSH doit être systématique avant la mise sous amiodarone. Il est suffisant pour confirmer l’euthyroï-
EMS Pharmacology Reference Guide - Rhode Island
health.ri.gov2. Amiodarone should not be used in individuals with polymorphic VT associated with a prolonged QT interval. 3. Amiodarone for infusion should only be diluted with D5W and given with an in-line filter. 4. Use with beta or calcium blocking agents may increase risk of hypotension and bradycardia. Significant adverse/side effects: 1. Hypotension
HIGHLIGHTS OF PRESCRIBING INFORMATION 4, 5, …
www.gilead.com• Coadministration with amiodarone may result in serious symptomatic bradycardia. Use of HARVONI with amiodarone is not recommended. (5.2, 6.2, 7.2)
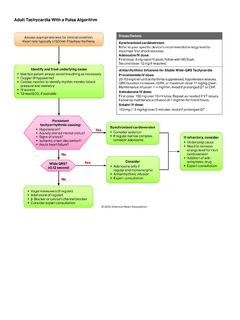
Adult Tachycardia With a Pulse Algorithm
cpr.heart.orgMaintenance infusion: 1-4 mg/min. Avoid if prolonged QT or CHF. Amiodarone IV dose: First dose: 150 mg over 10 minutes. Repeat as needed if VT recurs. Follow by maintenance infusion of 1 mg/min for first 6 hours. Sotalol IV dose: 100 mg (1.5 mg/kg) over 5 minutes. Avoid if prolonged QT. Persistent tachyarrhythmia causing: • Hypotension?
Attach Patient Label Here Kg - SORT
www.sort.nhs.ukWEIGHT Date Emergency. Cardiovascular. Adrenaline 1:10,000 Atropine 600mcg/ml Sodium Bicarbonate 8.4% Calcium Gluconate 10% Adenosine Amiodarone Load Cardioversion (sync)
Table 1: Medications that influence warfarin therapy
www.silkview.comMedications that influence warfarin therapy Significant increase in the INR Significant decrease in the INR Amiodarone Amobarbital Anabolic steroids Aprepitant Bactrim (TMP/SMZ) Butabarbital Chloramphenicol Carbamazepine Disulfiram Dicloxacillin Fluconazole Griseofulvin Isoniazid Methimazole Metronidazole Phenobarbital …
List of IP Reference Substances available at IPC, Ghaziabad
chromachemie.co.in17. Aminophylline IPRS/43/11 200 mg 18. Amiodarone Hydrochloride IPRS/45/11 200 mg 19. Amisulpride IPRS/69/13 200 mg 20. Amitriptyline Hydrochloride IPRS/31/15 IPRS/20/11 (05/2016) 200 mg 21. Amlodipine Besylate IPRS/66/14 200 mg 22. Amodiaquine Hydrochloride IPRS/65/10 200 mg 23. Amoxycillin Sodium IPRS/218/12 200 mg 24.
Specials and expensive liquids guideline
www.derbyshiremedicinesmanagement.nhs.ukAmiodarone The tablets can be crushed and mixed with water for administration. Without crushing they disperse in around five minutes. Give immediately. The crushed tablets have a bitter taste, for swallowing difficulties they can be mixed with fruit juice if desired.1 UNLICENSED 50mg/5ml and 100mg/5ml oral suspension included
Critical Care Intravenous Drug Administration Guide - SGUL
www.gicu.sgul.ac.ukAmiodarone Continuous or Intermittent infusion with pump preferably by central line Loading dose (5mg/kg in 250ml G) by intermittent infusion over 20 to 120 minutes, followed by continuous infusion (up to 15mg/kg in up to 500ml G over 24 hours) Dilute in G only. Stability concentration dependent - do not dilute to less than 300mg in 500mls. Usual
UKCPA Minimum Volumes Guide 2012
scottishintensivecare.org.ukUKCPA: Minimum Infusion Volumes for Fluid Restricted Critically Ill Patients 4th Edition (v4.4) December 2012 Page | 8 United Kingdom Clinical Pharmacy Association C ritical C are G roup Amiodarone Data Sheet Recommendations From 150mg in 250ml of glucose 5% (0.6mg/ml) up to 1.2g in 500ml of glucose 5% (2.4mg/ml) .
Resuscitation trolley checklist - Queensland Health
www.health.qld.gov.auAmiodarone ampoule 150 mg/3 mL x 3 Aspirin 300 mg dispersible tablet sleeve x 1 Atropine sulphate 600 microg/mL ampoule x 2 Glyceryl trinitrate tablets 600 microg bottle x 1 Second Line Adenosine 6 mg/2 mL ampoule x 6 Adrenaline 1:1000 ampoule x 10 Calcium gluconate 2.2mmol of calcium in 10mL vial x 4 Ceftriaxone 1 g vial x 2 Droperidol 10 mg/2 ...
The Renal Drug Handbook - SGUL
www.gicu.sgul.ac.ukAmiodarone hydrochloride 39 Amisulpride 41 Amitriptyline hydrochloride 42 Amlodipine 44 Amoxicillin 45 Amphotericin iV – Abelcet (lipid complex) 46 Amphotericin iV – Ambisome (liposomal) 47 Amphotericin iV – Amphocil (complex with sodium cholesteryl sulphate) 49 Amphotericin iV – Fungizone 50
Pediatric Guidelines for IV Medication Administration
www.umassmed.eduInfusion Concen- tration Usual Dosing and Administration Comments Amiodarone X (Cordarone®) exceeding c X Bolus in ode only No infusion X Bolus diluted to 1.5 -3 mg/mLin D5W Infusion 450 mg/ 250 mL in D5W BOLUS: PALS for pulseless VF/VT5 mg/kg (MAX 300 mg/dose) given over 5 10 minutes. 0.22 micron filter preferred . Flush post dose.
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS
www.ema.europa.euEach hard capsule contains ozanimod hydrochloride equivalent to 0.23 mg ozanimod. Zeposia 0.46 mg hard capsules ... or class III (e.g. amiodarone, sotalol) antiarrhythmic medicinal products, which have been associated with cases of torsades de pointes in patients with bradycardia have not been studied with ozanimod. Liver function
ตารางการบริหารยาและความคงตัวของยาฉีดภายหลังผสม
www.nbhospital.go.thAmiodarone HCl (Cordarone®) : 150 mg/3 ml IV push มากกวา 3 min (central line) - - - D5W ขึ นอยูกับ ภาชนะที่ใช a - - เก็บพนแสง - สารละลายบรรจุในภาชนะพลาสติก pvc
Philippine National
caro.doh.gov.phInj.: 25 mg/mL, 10 mL ampul (IV) Amiodarone (as hydrochloride) (2) Oral: 200 mg tablet Inj.: 50 mg/mL, 3 mL ampul (IV) (2) Amlodipine (as besilate/camsylate) Oral: 5 mg and 10 mg tablet Amoxicillin (as trihydrate) Oral: 250 mg and 500 mg capsule 100 mg/mL granules/powder for drops (suspension), 15 mL
The Society of Thoracic Surgeons Practice …
www.sts.org(versus 0% in the placebo group). Among pneumonec-tomy patients, the ARDS rate was 27%. Amiodarone-induced pulmonary toxicity appears to occur not only after chronic administration, but also
Amiodarone 引發㆙狀腺毒症 - tsim.org.tw
www.tsim.org.tw3 應密切監測㆙狀腺功能,以作為調整amiodarone 劑量的參考。如病㆟在使用amiodarone 後 有FT4 持續升高,TSH 數據偏低的情形,則顯示可能有㆙狀腺高能症的情形,而㆙狀腺抗體 的正常與否則端視病㆟用藥時間的長短,㆙狀腺抗體呈陰性反應可能是因為amiodarone 使
Amiodarone Hydrochloride 150 mg/3 mL Name of the …
www.medsafe.govt.nz5 Amiodarone Hydrochloride 150 mg/3 mL 01102015 Thyroid hormone abnormalities As amiodarone may induce thyroid disorders, particularly in patients with personal or family
Similar queries
Amiodarone, Infusion, PRESCRIBING INFORMATION, Tachycardia, Attach Patient Label Here Kg, Medications that influence warfarin therapy, IP Reference Substances available at IPC, Ghaziabad, Amiodarone hydrochloride, Hydrochloride, Specials and expensive liquids guideline, Critical Care Intravenous Drug Administration Guide, UKCPA Minimum, UKCPA: Minimum Infusion, Resuscitation trolley checklist, 150 mg, Pediatric Guidelines for IV Medication Administration, The Society of Thoracic Surgeons, Induced, Toxicity, Amiodarone Hydrochloride 150 mg/3, Thyroid