CDSCO - Guidance for Industry
4.8.3 Certificate of analysis (Pilot scale batches) 4.9. Reference standards 4.10. Container closure system 4.10.1 Packaging Materials: Specifications and Test methods 4.10.2 Art work – Packaging material (label, primary carton, secondary carton and Pack Insert. 4.10.3 Packaging Specifications 4.11 Stability data
Tags:
Information
Domain:
Source:
Link to this page:
Please notify us if you found a problem with this document:
Documents from same domain
CENTRAL DRUGS STANDARD CONTROL ORGANIZATION …
cdsco.gov.in1. Application in Form-44 duly signed, by the Authorized signatory with details of name and designation along with relevant drugs details and utilization. 2. Treasury Challan (TR–6)/Bharatkosh receipt of Rs. 25000/- (Medical and Public Health Account: 0210). 3. Application in Form-12 duly signed, by the Authorized signatory with details
SUSPECTED ADVERSE DRUG REACTION REPORTING FORM
cdsco.gov.inDuly filled Suspected Adverse Drug Reaction Reporting Form can be send to the nearest Adverse Drug Reaction Monitoring Centre (AMC) or directly to the National Coordination Centre (NCC). Call on Helpline (Toll Free) 1800 180 3024 to report ADRs.
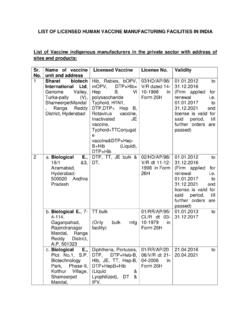
LIST OF LICENSED HUMAN VACCINE MANUFACTURING …
cdsco.gov.in8 Green signal BioPharma Ltd, 49, Pappankuppan Village, Gummidipoondi, Chennai–601201 BCG Vaccine TN00002462 in Form 28D 12.05.2016 to 11.05.2021 9 a. Panacea,
GUIDELINES ON RECALL AND RAPID ALERT SYSTEM FOR …
cdsco.gov.inThe defective products related to quality includes Not of Standard Quality, Adulterated or Spurious drugs. Safety and efficacy related recalls include serious adverse reactions and death. Recalls also include drugs prohibited under the Provisions of Drugs & Cosmetics Act and also those products for which product licenses are suspended/cancelled.
Guidelines, System, Quality, Safety, Alert, Recall, Cosmetic, Rapid, Efficacy, Guidelines on recall and rapid alert system for
1. APPLICATIONS FOR INVESTIGATIONAL NEW DRUGS …
cdsco.gov.in2 Name of the applicant 3 Name of the New Drug a. Composition of the New Drug b. Dosage Form c. Proposed indication for the New Drug 4 Application in Form 44 complete in all respect duly signed and stamped by authorized person of the firm 5 Treasury Challan of INR 50,000 (for Phase I) or INR 25,000 (for Phase II / III)
Applications, Drug, Investigational, Investigational new drug applications for, New drugs
KM 554e-20180422102116 - CDSCO
cdsco.gov.in(vi) "Hazard" potential source of harm (vii) "Lay person" means an individual that does not have formal training in a relevant field or discipline. (viii)"Risk" means combination of the probability of occurrence of harm and the severity of that harm. (ix) Words and expressions used but not defined in these guidelines shall have the
National List of Essential Medicines (NLEM) 2015 - CDSCO
cdsco.gov.inNational List of Essential Medicines (NLEM) 2015 Page 6 of 38 The Core-Committee through a series of meetings and consultations across the country, deliberated and revised the National List of Essential Medicines
Related documents
GOVERNMENT OF MAHARASHTRA
view.mahacet.orgIndian Origin (PIO) cardholders registered under notification of the ... “SSC” means the Secondary School Certificate (Standard X) examination conducted by Maharashtra State Board of Secondary and Higher Secondary ... rules and regulations made there under. (4) The Candidates seeking admissions to the professional courses for the seats
2022 Summary of Benefits
www.nmrhca.orgDavis Vision 1-800-999-5431 6301 Indian School Rd NE, Ste 200 All prospective clients can use code 7587 when Albuquerque, NM 87110 ... provisions of the Retiree Health Care Act (Act) or NMRHCA Rules and Regulations (which can be found on the NMRHCA website: www.nmrhca.org) ... NMRHCA program of health care benefits be secondary to your …
CKYC & KRA KYC Form LOGO Know Your Client Application …
www.mfuindia.com11.For opening an account with Depository participant or Mutual Fund, for a minor, photocopy of the School Leaving Certificate/Mark sheet issued by Higher Secondary Board/ Passport of Minor/Birth Certificate must be provided. A. Clarification / Guidelines on …
CKYC & KRA KYC Form
www.utimf.com11.For opening an account with Depository participant or Mutual Fund, for a minor, photocopy of the School Leaving Certificate/Mark sheet issued by Higher Secondary Board/ Passport of Minor/Birth Certificate must be provided. A. Clari˜cation / Guidelines on …
FACULTY OF EDUCATION
unipune.ac.in(Based on NCTE Regulations 2014) B.Ed. FIRST YEAR Course Code Title of the Course Credits Hours Marks (External +Internal) PERSPECTIVES OF EDUCATION – CORE COURSES BED101 Childhood and Growing up 4 64 80+20 BED102 Contemporary Indian Education, Gender and Society 4 64 80+20 BED103 Learning and Teaching 4 64 80+20