The COVID-19 Treatment Guidelines Panel’s Interim ...
Dec 23, 2021 · response to COVID-19 vaccination. Unlike anti-SARS-CoV-2 agents used for treatment, tixagevimab plus cilgavimab (Evusheld) is not authorized for use in unvaccinated individuals unless full vaccination is not possible due to a history of severe allergic reaction to the COVID-19 vaccine. Generally speaking,
Tags:
Treatment, Cargille, Reactions, Severe, Of severe allergic reaction
Information
Domain:
Source:
Link to this page:
Please notify us if you found a problem with this document:
Documents from same domain
The COVID-19 Treatment Guidelines Panel’s Statement on the ...
files.covid19treatmentguidelines.nih.govJul 08, 2021 · Panel Recommendations • The quality of the data that supports the recommendations for the use of anti-SARS-CoV-2 monoclonal antibodies differs based on the criteria for high risk of progression to severe COVID-19 used. Consequently, the Panel weighed the strength of the recommendations based on the evidence for the risk of progression.
Coronavirus Disease 2019 (COVID-19) Treatment Guidelines
files.covid19treatmentguidelines.nih.govCoronavirus Disease 2019 (COVID-19) Treatment Guidelines. is published in an electronic format that can be updated in step with the rapid pace and growing volume of information regarding the treatment of COVID-19. The COVID-19 Treatment Guidelines Panel (the Panel) is committed to updating this document to
Treatment, Disease, Coronavirus, Covid, Covid 19, Coronavirus disease, Covid 19 treatment
Antiviral Drugs That Are Approved or Under Evaluation …
files.covid19treatmentguidelines.nih.govCOVID-19 Treatment Guidelines 99 Antiviral Drugs That Are Approved or Under Evaluation for . the Treatment of COVID-19 . Last Updated: December 16, 2021. Summary Recommendations. Remdesivir is the only drug that is approved by the Food and Drug Administration for the treatment of COVID-19. In
Evaluation, Drug, Treatment, Under, That, Approved, Antiviral, The treatment, Antiviral drugs that are approved or under evaluation, Antiviral drugs that are approved or under evaluation for
Antiviral Drugs That Are Approved or Under Evaluation …
files.covid19treatmentguidelines.nih.govRemdesivir is the only drug that is approved by the Food and Drug Administration for the treatment of COVID-19. In . this section, the COVID-19 Treatment Guidelines Panel (the Panel) provides recommendations for using antiviral drugs to . treat COVID-19 based on the available data. For more information on these antiviral agents, see . Table 2f ...
Guidelines, Evaluation, Drug, Under, That, Approved, Antiviral, Antiviral drugs that are approved or under evaluation
What’s New in the Guidelines
files.covid19treatmentguidelines.nih.govDec 23, 2021 · This section has been updated to incorporate results from the REMDACTA trial, a double-blind, placebo-controlled, randomized trial that evaluated the use of tocilizumab in combination with remdesivir in patients who were hospitalized with severe COVID-19 pneumonia. Kinase Inhibitors: Janus Kinase Inhibitors and Bruton’s Tyrosine Kinase Inhibitors
Last Updated: December 16, 2021 - National Institutes of ...
files.covid19treatmentguidelines.nih.govDec 16, 2021 · recommends prone ventilation for 12 to 16 hours per day over no prone ventilation ... Severe cases of COVID-19 may be associated with hypoxemic respiratory failure, acute respiratory . distress syndrome (ARDS), septic shock, cardiac dysfunction, elevation in multiple inflammatory .
Ventilation, Respiratory, Acute, Enpro, Distress, Acute respiratory, Covid, Prone ventilation
The COVID-19 Treatment Guidelines Panel’s Statement on ...
files.covid19treatmentguidelines.nih.govJan 05, 2022 · with worse clinical outcomes.3,4 Hospitalized patients with COVID-19 are at high risk for venous thromboembolism (VTE).5 At a minimum, hospitalized COVID-19 patients should receive prophylactic doses of anticoagulants, such as low molecular weight heparin (LMWH) or unfractionated heparin, for the duration of their hospitalization. Recommendations
Epidemiology - National Institutes of Health
files.covid19treatmentguidelines.nih.govDec 16, 2021 · Like other RNA viruses, SARS-CoV-2 is constantly evolving through random mutations. New mutations can potentially increase or decrease infectiousness and virulence. In addition, mutations can increase the ... being monitored (VBM). This refers to variants for which the data indicate a potential or clear impact on
Last Updated: December 16, 2021 - National Institutes of ...
files.covid19treatmentguidelines.nih.govDec 16, 2021 · Anti-SARS-CoV-2 Monoclonal Antibodies Last Updated: December 16, 2021 The SARS-CoV-2 genome encodes 4 major structural proteins: spike (S), envelope (E), membrane (M), and nucleocapsid (N), as well as nonstructural and accessory proteins. The spike protein is further divided into 2 subunits, S1 and S2, that mediate host cell attachment and ...
Antiviral Drugs That Are Approved or Under Evaluation for ...
files.covid19treatmentguidelines.nih.govRemdesivir is a nucleotide prodrug of an adenosine analog. It binds to the viral RNA-dependent RNA . polymerase and inhibits viral replication by terminating RNA transcription prematurely. Remdesivir . has demonstrated in vitro activity against SARS-CoV-2. 1. In a rhesus macaque model of SARS-CoV-2
Related documents
DENTAL TREATMENT CONSENT FORM
www.arlingtondentalgrp.commedications can cause allergic reactions causing redness and swelling of tissues, pain, itching, vomiting, and/or anaphylactic shock (severe allergic reaction). (Initials_____) 3. CHANGES IN TREATMENT PLAN I understand that during treatment it may be necessary to change or add procedures because of conditions found while working on
Treatment, Cargille, Reactions, Severe, Severe allergic reactions
Acute and Chronic Urticaria: Evaluation and Treatment
www.aafp.orgJun 01, 2017 · gic reaction. Urticaria is caused by immunoglobulin E- and non–immunoglobulin E-mediated release of histamine ... treatment in severe allergic reactions. American Academy of Allergy, Asthma, and ...
TREATMENT OF SEVERE ALLERGIC REACTION
www.co.washington.or.usTREATMENT OF SEVERE ALLERGIC REACTION: A Protocol for Training. 1. I. INTRODUCTION. Anaphylaxis is a severe, potentially fatal allergic reaction. It is characteristically unexpected and rapid in onset. Immediate injection . of epinephrine is the single factor most likely to save a life under these circumstances. It is estimated that between one ...
Treatment, Cargille, Reactions, Severe, Allergic reaction, Treatment of severe allergic reaction
What is immunoglobulin treatment? What are the side effects?
www.blood.gov.auof treatment, or for treatment over a longer period. ... particles or because of a reaction that you had and did not use the entire product. ... 6 headache 6 nausea (feeling sick) 6 stomach and back pain Rarely, more serious allergic reactions occur. Symptoms include: 6 shortness of breath 6 wheezing 6 tongue/throat swelling 6 chest tightness 6 ...
Emergency treatment of anaphylactic reactions
www.resus.org.uk– A severe, life-threatening, generalized or systemic hypersensitivity reaction Anaphylaxis is characterised by: – Rapidly developing, life threatening, Airway and/or Breathing and or Circulation problems – Usually with skin and/or mucosal changes
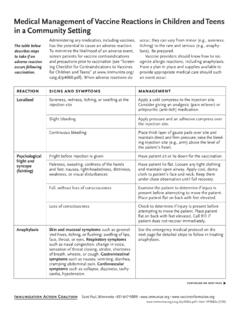
Medical Management of Vaccine Reactions in Children and ...
www.immunize.orgognize allergic reactions, including anaphylaxis. Have a plan in place and supplies available to provide appropriate medical care should such an event occur. The table below. describes steps to take if an . adverse reaction occurs following vaccination. reaction