Pharmaceutical Analysis Using UV-Vis: Compliance with USP …
standard deviation for each analysis wavelength. Control of Wavelength Recommended Reference Material Holmium in perchloric acid solution. 200 – 400 nm ± 1 nm 400 – 780 nm ± 2 nm ≤ 0.5 nm standard deviation Cerium sulfate solution 200 – 400 nm ± 1 nm ≤ 0.5 nm standard deviation Didymium solution 400 – 900 nm ± 2 nm
Download Pharmaceutical Analysis Using UV-Vis: Compliance with USP …
Information
Domain:
Source:
Link to this page:
Please notify us if you found a problem with this document:
Documents from same domain
Agilent 7820A Gas Chromatograph
www.agilent.comAdvanced User Guide 7 Controlling a V 115 From the keyboard 115 From the run or clock time tables 115 Gas sampling valve 115
Guidelines for Trouble Shooting and Maintenance …
www.agilent.comGuidelines for Trouble Shooting and Maintenance of ICP-OES Systems Presented by Eric Vanclay, Spectroscopy Supplies Marketing Manager ICP-OES Maintenance & Trouble Shooting Nov. 2016
Maintenance, Shooting, Trouble, For trouble shooting and maintenance, For trouble shooting and maintenance of
Unraveling the Complexity of Lipidomes by ... - Agilent
www.agilent.com3 Software • Agilent OpenLAB CDS ChemStation Edition (revision C.01.07) with Agilent 1290 Infi nity 2D-LC Software (revision A.01.02) • Agilent MassHunter for instrument
Agilent OpenLAB Chromatography Data System …
www.agilent.comContents . 4 Supported Instruments and Firmware Guide . GC Sampler, Autosampler and Tray Compatibility 19 6850 ALS hardware 20 7650 ALS hardware: 20
System, Data, Chromatography, Agilent, Openlab, Agilent openlab chromatography data system
Quantitative Analysis of Underivatized Amino Acids …
www.agilent.com2 Introduction A wide range of amino acids are present in plant material at parts-per-million (ppm) and parts-per-billion (ppb) levels. As previously found1, underivatized amino acids can be analyzed with excellent resolution and sensitivity using
Certificate of Analysis - Agilent
www.agilent.comCertificate of Analysis . Headspace OQ/PV Standard Kit . Agilent Part Sample Lot . Number: 5182-9733 Number: N082208
Support your lab’s evolving storage requirements
www.agilent.comSupport your lab’s evolving storage requirements Care-free maintenance with industry-standard infrastructure Adding new platforms typically results in more work for the IT
Page 1/8 Safety Data Sheet acc. to OSHA HCS 1 …
www.agilent.comPage 2/8 Safety Data Sheet acc. to OSHA HCS Printing date 04/28/2016 Reviewed on 04/28/2016 Product Name: Tuning Solution for ICP-MS, Part Number 5185-5959
Sheet, Data, Safety, Pages, Osha, 8 safety data sheet acc, To osha hcs, Page 1 8 safety data sheet acc, To osha hcs 1
Introduction to Quantitative PCR - Agilent
www.agilent.comIntroduction to Quantitative PCR Methods and Applications Guide IN 70200 D US and Canada Orders: 800-227-9770 x3 Technical Service: 800-227-9770 x2
Introduction, Methods, Quantitative, Introduction to quantitative pcr, Introduction to quantitative pcr methods
Herceptest™ Interpretation Manual - Breast Cancer
www.agilent.com5. Introduction 6. HER2 Overview 6 . HER2 Protein and HER2 Family 6 . HER2 Testing IHC and FISH 7 . HER2 Testing Algorithm 8 . The HercepTest TM Kit 9
Manual, Testing, Overview, Interpretation, Breast, Cancer, Interpretation manual breast cancer
Related documents
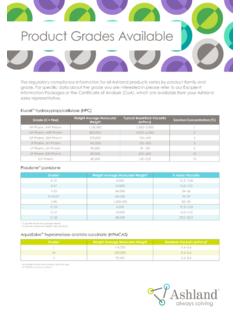
Product Grades Available - Ashland
www.ashland.comInformation Packages or the Certificate of Analysis (CoA), which are available from your Ashland sales representative. Klucel™ hydroxypropylcellulose (HPC) Grade (X = Fine) Weight Average Molecular Weight Typical Brookfield Viscosity (mPa•s) Solution Concentration (%) HF Pharm, HXF Pharm 1,150,000 1,500–3,000 1
2021-09-08-Report on pharmaceutical aspects on impact of …
www.ema.europa.euprovide an analysis with the aim to define the technical purpose of titanium dioxide in medicinal products; feasibility of alternatives to replace it without negative impacting the quality, safety and efficacy of medicines; and if confirmed, considerations to be taken into account to define a transition period for phasing out this excipient.
<1231> WATER FOR PHARMACEUTICAL PURPOSES
www.drugfuture.comFirst Supplement to USP 35–NF 30 General Information / 〈1231〉 Water for Pharmaceutical Purposes5219 incident on the sample and includes losses due to solvent nature of this raw material. Microbial specifications are typi-absorption, refraction, and scattering; and A is the cally assessed by test methods that take at least 48 to 72
6 Ways Pharmaceutical Companies are Using Data Analytics …
www.iqpc.com6 Ways Pharmaceutical Companies are Using Big Data to Drive Innovation & Value . Page | 3 #1: Accelerate drug discovery and development . With a large number of patents for blockbuster drugs expired or near expiration and the cost of bringing a new drug to market pushing $5 billion, according to a 2013 Forbes analysis, there are
ICH Q10 Pharmaceutical Quality System
database.ich.orgPharmaceutical Quality System 2. Management Responsibility 3. Continual Improvement of Process Performance and Product Quality 4. Continual Improvement of the Pharmaceutical Quality System 5. Glossary Annex 1 - Potential Opportunities to Enhance Science and Risk Based Regulatory Approaches
Pharmaceutical regulation in 15 European countries
www.euro.who.intPharmaceutical regulation in 15 European countries Review Vol. 18 No. 5 2016 Health Systems in Transition Dimitra Panteli • Francis Arickx • Irina Cleemput Guillaume Dedet • Helene Eckhardt • Emer Fogarty Sophie Gerkens • Cornelia Henschke • Jennifer Hislop Claudio Jommi • Daphne Kaitelidou • Pawel Kawalec Ilmo Keskimäki • Madelon Kroneman • Julio Lopez Bastida