Amines
Found 10 free book(s)Chapter 17: Amines and Amides - latech.edu
www.chem.latech.eduAmines with fewer than six carbon atoms are infinitely soluble in water. Solubility results from hydrogen bonding between the amines and water. Even tertiary amines are water-soluble because of its ability to form hydrogen bonds. Odor or smell Methylamines (mono-, di-, and tri-) and ethylamine (gases) have ammonia-like smell.
Ch 06 Amines and Amides - Angelo State University
www.angelo.edu• Amines in the IUPAC system: the “e” ending of the alkane name for the longest chain is replaced with –amine. The amine group is located by the position number. Groups that are attached to the nitrogen atom are located using “N” as the position number. More complex primary amines are named
Classification and Nomenclature of Amines
www.angelo.edu• Amines in the IUPAC system: the “e” ending of the alkane name for the longest chain is replaced with –amine. The amine group is located by the position number. Groups that are attached to the nitrogen atom are located using “N” as the position number. More complex primary amines are named
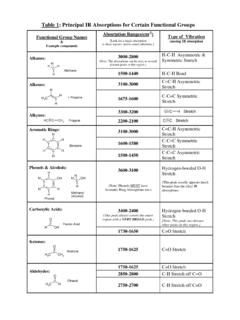
Table 1: Principal IR Absorptions for Certain Functional ...
academics.wellesley.eduAmines—Secondary: N-H Stretch N CH3 H N-Methylethylamine 1550-1450 N-H Bend Nitriles: C C N H H H Methanenitrile 2300-2200 CNStretch 1600-1500 N=O Stretch Nitro Groups: H C 3 N + O O Nitromethane (Note: Both peaks are <200 cm-1 apart.) 1400-1300 N=O Bend 3500-3100 N-H Stretch (similar to amines) 1670-1600 C=O Stretch Amides: H C 3 C NH 2 O ...
UE1 : Biomolécules (1) : Acides aminés et protéines ...
unf3s.cerimes.frdécarboxylation : synthèse amines biogènes (ex : histamine) Réactions du NH 2 N-alkylation : méthylés (ex: bétaïne) N-arylation : analyse ( DNP avec fluorodinitrobenzéne) N-acylation : synthèse peptidique Analyse Anhydride, halogénure Formation de bases de Schiff : réaction avec aldéhyde Désamination, transamination réaction globale
Synthesis of Carboxylic Acids
web.mnstate.eduNaOH (or other bases, including amines) Na carboxylate salt (basic) • Mechanism: Required (deprotonation) • Reverse Mechanism: Required (protonation) • Carboxylic acids are completely converted to carboxylate salts by base • Carboxylate salts are completely neutralized back to carboxylic acids by strong acid
Chemical Classes of Opioids (Updated 10/1/2018 ...
opioidcalculator.practicalpainmanagement.comAMINES MORPHINE PENTAZOCINE FENTANYL METHADONE TRAMADOL Buprenorphine* Butorphanol* Codeine Dextromethorphan* Dihydrocodeine Heroin (diacetyl-morphine) Hydrocodone* Hydromorphone* Levorphanol* Methylnaltrexone** Morphine (Opium, conc) Nalbuphine* Naloxone* Naloxegol* Naltrexone** Oxycodone*
Functional Groups - Purdue University
www.chem.purdue.eduG. Amines • contain an “amino” group – a N atom bonded to 1, 2, or 3 carbon atom groups by single bonds H. Aldehydes and Ketones • contain a C=O (“carbonyl”) group • note that in condensed structural formulas, the aldehyde group may be written as –CH=O or as –CHO I. Carboxylic Acids
TECHNIQUES: Principes de la chromatographie
zysman-colman.com6 11 Deux facteurs interviennent lors de l’interaction entre l’ éluant et le soluté (mélange de composés à séparer): • la solubilité: on doit être en mesure de dissoudre le soluté dans l’éluant pour que la migration se fasse. • la polarité de l’éluant va déterminer à quelle vitesse le composé migre. Moins un composé est polaire, moins il s’accroche à l ...
Consider the following reaction sequence starting from ...
drwainwright.weebly.com(c) €€€€The amine CH3CH2CH2NH2 can be prepared by two different routes. Route A is a two-stage process and starts from CH3CH2Br. Route B is a one-stage process and starts from CH3CH2CH2Br. (i)€€€€€€Identify the intermediate compound in Route A.