Atomic Masses
Found 9 free book(s)Example Exercise 9.1 Atomic Mass and Avogadro’s Number
www.austincc.eduWe begin by finding the atomic mass of each element in the periodic table. The molar mass equals the sum of the atomic masses expressed in g/mol. (a) The atomic mass of Ag is 107.87 amu, and the molar mass of silver equals 107.87 g/mol. (b) The sum of the atomic masses for NH. 3 is 14.01 amu + 3(1.01)amu = 17.04 amu. The molar mass of ammonia
Average Atomic Mass
mrsaultclassroom.weebly.comof atomic mass (and for unstable isotopes, radioactivity). Therefore, the periodic table lists a weighted average atomic mass for each element. In order to calculate this quantity, the natural abundance and atomic mass of each isotope must be provided. •N16. Consider the individual atomic masses for magnesium isotopes given in Model 2. a.
Nuclear Masses and Mass Excess: Q values for Nuclear …
www.astro.princeton.edumass of one 4He atom is about 0.71% less than the combined rest masses of four hydrogen atoms (note that the electrons are included in the atomic masses here). The difference, or about 26.7MeV, is released as heat, except for ≈ 0.6MeV worth of neutrinos (in the ppchain). There are two paths from 41Hto 4He: the pp cycle, which predominates in
Atomic mass is based on a relative scale and the mass of ...
www.westfield.ma.eduatomic masses stays the same. Since atoms of C are so small, we could place enough of them on a balance so that the mass would be 12.01 g. The same could be done with W; that is, 183.9 g of W could be placed on a balance. mass of W = 183.9 g = 15.31 mass of C 12.01 g 1 Since the ratio of the masses is the same, each sample contains 28
NAME Average Atomic Mass Worksheet: show all work.
www.npsd.k12.nj.usThe masses on the periodic table are the average mass of all isotopes and their abundances found in the universe. Although carbon-12 weighs exactly 12 amu, the ... atomic mass of 62.9298 amu and an abundance of 69.09%. The other isotope, 65Cu, has an abundance of 30.91%. The average atomic mass between these two isotopes is 63.546 amu.
Basic Atomic Structure Worksheet Key 1
www.neshaminy.orgThe atomic number gives the "identity" of an element as well as its location on the periodic table. No two different elements will have the atomic number. The mQSS of an element is the average mass of an element's naturally occurring atom, or isotopes, taking …
Table of Isotopic Masses and Natural Abundances
www.chem.ualberta.caTable of Isotopic Masses and Natural Abundances This table lists the mass and percent natural abundance for the stable nuclides. The mass of the longest lived isotope is given for elements without a stable nuclide. Nuclides marked with an asterisk (*) in the abundance column
Isotope Worksheet Answer Key - ISD 622
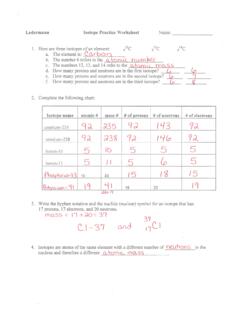
www.isd622.orgatomic # Phos hocus-33 15 Write the hyphen notation and the nuclide (nuclear) symbol for an isotope that has 17 protons, 17 electrons, and 20 neutrons. 37 Isotopes are atoms of the same element with a different number of nucleus and therefore a different DA OSS in the . Ledermann
Atomic Mass and Atomic Number Worksheet Key
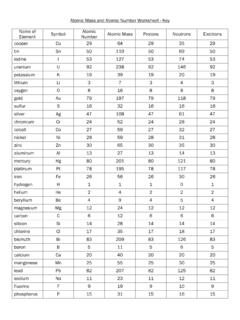
www.readington.k12.nj.usAtomic Mass and Atomic Number Worksheet - Key Name of Element Symbol Atomic Number Atomic Mass Protons Neutrons Electrons copper Cu 29 64 29 35 29 tin Sn 50 119 50 69 50 iodine I 53 127 53 74 53 uranium U 92 238 92 146 92 potassium K 19 39 19 20 19 lithium Li 3 7 3 4 3 oxygen O 8 16 8 8 8 ...