Isotope practice
Found 10 free book(s)Isotope Worksheet Answer Key - ISD 622
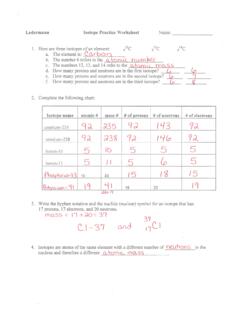
www.isd622.orgIsotope Practice Worksheet Name: 1. 2. D. 4. 13 12 Here are three isotopes of an element: a. The element is: b. The number 6 refers to the c. The numbers 12, 13, and 14 refer to the d. How many protons and neutrons are in the first isotope? (O e. How many protons and neutrons are in the second isotope? f.
Nuclear Chemistry Practice Problems - Utah State University
ion.chem.usu.eduChemistry 1110 – Chapter 5 – Nuclear Chemistry – Practice Problems Page | 10 47. The half-life of a radioisotope is A) one-half of the time it takes for the radioisotope to completely decay to a nonradioactive isotope. B) the time it takes for the radioisotope to become an isotope with one-half of the atomic
half-life practice questions - Loudoun County Public Schools
www.lcps.orghalf-life practice questions 1. An 80 milligram sample of a radioactive isotope decays to 5 milligrams in 32 days. What is the half-life of this element? A. 8 days B. 2 days C. 16 days D. 4 days 1. 2. An original sample of a radioisotope had a mass of 10 grams. After 2 days, 5 grams of the radioisotope remains unchanged.
isotopic abundance practice problems - Maurer Math
maurermath.weebly.com! isotopic abundance - practice problems The atomic mass for each element appearing on the periodic table represents the weighted average of masses for each individual isotope of an element. For example, the atomic mass of carbon is reported as 12.011 amu (atomic mass units). Carbon is composed primarily of two isotopes; carbon-12 and carbon-14.
Protons, Neutrons, and Electrons Practice Worksheet
www.mrphysics.orgProtons, Neutrons, and Electrons Practice Worksheet Fill in the blanks in the following worksheet. Please keep in mind that the isotope represented by each space may NOT be the most common isotope or the one closest in atomic mass to the value on the periodic table. Atomic symbol Atomic number Protons Neutrons Electrons Atomic mass B6 11 24 31 ...
Grade 11 Science Practice Test - Nebraska
www.education.ne.gov20. How does one isotope of an element differ from another isotope of the same element? A. increase or decrease in the charge B. increase or decrease in the number of protons C. increase or decrease in the number of neutrons D. increase or decrease in the number of electrons 635188 / I20 SCIENCE PRACTICE TEST SP12S11XP01 12 STOP.
Mass spectrometry practice questions - Weebly
www.sandsci.weebly.comMass spectrometry practice questions 157 ... €€€€ The table below gives the percentage abundance of each isotope in the mass spectrum of a sample of titanium. € Define the term relative atomic mass of an element. Use the above data to calculate the
Radioactivity and Balancing Nuclear Reactions: Balancing ...
web.mnstate.eduExtra Practice Problems Radioactivity and Balancing Nuclear Reactions: Balancing Nuclear Reactions and Understanding which Particles are Involved p1 Miscellaneous p9 The Stability of Atomic Nuclei: The Belt of Stability, Recognizing Whether An Isotope is likely to be stable or not, and predicting what it will do if it isn’t. p5 Mass Deficit. ...
Isotope Practice Worksheet - Sir Thomas Rich's School
strschool.co.ukIsotope Practice Worksheet 1. Here are three isotopes of an element: 6 12C 6 13C 6 14C a. The element is: Carbon b. The number 6 refers to the atomic number c. The numbers 12, 13, and 14 refer to the mass number d. How many protons and neutrons are in the first isotope? 12 e. How many protons and neutrons are in the second isotope? 13 f.
Isotope Practice Worksheet - Chemistry
atischemistry.weebly.comIsotope Practice 1. Here are three isotopes of an element: 12C 14 13C C a. The element is: Carbon b. The number 6 refers to the Atomic Number c. The numbers 12, 13, and 14 refer to the Mass Number d. How many protons and neutrons are in the first isotope? 6 protons & 6 neutrons e. How many protons and neutrons are in the second isotope?