Surface Tension Of Polymers
Found 11 free book(s)Understanding Rheology of Thermoplastic Polymers
www.tainstruments.comor surface smoothness in a variety of thermoplastic molded ... polymers is more shear rate dependent than is the viscosity ... such as the interfacial tension become important and can change the rheological signature of the blend significantly. Moreover, the elastic properties of non-compatible blends ...
Sol Mech course text Feb10 - Harvard University
esag.harvard.eduIf we consider a horizontal surface through the material of the rod, it will be evident that ... materials and products of our technological civ ilization, e.g., by extruding metals or polymers through dies, rolling material into sheets, punc hing out complex shapes, etc.? ... had investigated the breaking loads of rods in tension and concluded ...
Surface Tension of Polymers - MIT
web.mit.edu2. Surface tension of solid surfaces (γ s) Methods for determining the surface tension of solids a) By measuring the contact angle between the solid and liquid b) By determining critical surface tension ( ) according to Zisman (1964), with the assumption that c) By extrapolating surface tension data of polymer melts to room
Physical Chemistry of Surfaces - sciencenet.cn
image.sciencenet.cnA. Surface Thermodynamic Quantities for a Pure Substance 48 B. The Total Surface Energy, Es 51 C. The Effect of Curvature on Vapor Pressure and Surface Tension 53 D. The Effect of Pressure on Surface Tension 55 2. Structural and Theoretical Treatments of Liquid Interfaces 56 A. Further Development of the Thermodynamic Treatment of the Surface ...
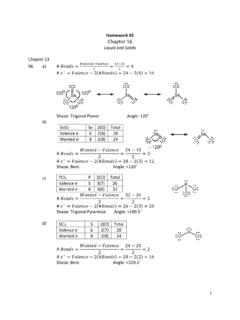
Homework #2 Chapter 16
people.chem.ucsb.edu27. a) Surface Tension: The tendency of molecules at the surface of a liquid to be pulled inward, resulting in a smooth surface. (The greater the intermolecular forces the greater the surface tension.) b) Viscosity: The resistance of a fluid (a gas or liquid) to flow: the higher the viscosity, the slower the flow.
urface tension alues o some common test liuids or surface ...
www.dataphysics-instruments.comurface tension alues o some common test liuids or surface energy analysis DataPhysics Instruments GmbH • phone + 49 (0)711 77 05 56 0 www.dataphysics-instruments.com • sales@dataphysics-instruments.com Name CAS Ref.-No. Surface tension @ 20 °C in mN/m Temperature coefficient in mN/(m K) 1,2-Dichloro ethane 107-06-2 33.30 -0.1428
Connective Tissue - Yale University
medcell.med.yale.eduCollagen is the main component that resist tension. Elastin also resist tension but behaves similar to rubber in that it can be stretched and will recoil after the force is removed. On the other side are glycosaminoglycans that resist compressive forces. Glycosaminoglycans are long sugar polymers that occupy large volumes within connective tissue.
MANUFACTURING PROPERTIES of ENGINEERING MATERIALS …
www2.isikun.edu.trPolymers are approximately 20% stronger in compression than in tension. In Ceramics, compressive strength is governed by crushing and is much larger than the tensile strength. Composites that contain fibers (including natural composites like wood) are a little weaker (up to 30%) in compression than tension as the fibers buckle
Importance, Objectives & Factors Affecting Dissolution ...
pharmaquest.weebly.cominterfacial tension, increasing effective surface area, which in turn results in faster dissolution rate. E.g Non-ionic surfactant Polysorbate 80 increase dissolution rate of phenacetin granules. The increase was more pronounced when the surfactant was sprayed on
1 Emulsion Formation, Stability, and Rheology
application.wiley-vch.detension γ. The surface free energy dGσ is made of three components: an entropy term Sσ dT, an interfacial energy term Adγ, and a composition term n idμ i (n i is the number of moles of component i with chemical potential μ i). The Gibbs–Deuhem equation is dGσ =−SσdT +Adγ + n idμ i (1.1) At constant temperature and composition dGσ ...
Klucel hydroxypropylcellulose - Ashland
www.ashland.comMost soluble polymers have a tendency to agglomerate, or lump, when the dry powder is first wet with solvent. Hydration of the outer surface of a particle, or an agglomeration of particles (lump), results in the formation of a viscous gel layer that inhibits wetting of …