Search results with tag "Lateral flow"
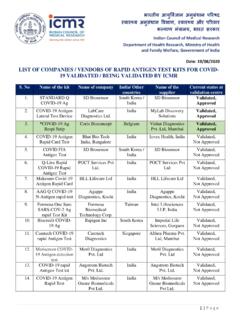
LIST OF COMPANIES / VENDORS OF RAPID ANTIGEN TEST …
stopcorona.tn.gov.inAntigen Lateral test Device (Lab Care Diagnostics Ltd) ... to be added to the well of the lateral flow strip. Read results in 15 mins or earlier after insertion of strip into tube containing sample and dilution buffer. The strip should be discarded after 15 minutes.
A guide for healthcare staff self-testing for coronavirus ...
www.england.nhs.ukLateral Flow Device (LFD) This guide will help you remain well at work and keep your patients safe. ... Ensure that the test device (called ‘antigen test cartridge’), swab and extraction solution are not damaged, broken or out of date. Use the test kit checklist to make
Panbio™ COVID-19 Antigen
www.tga.gov.au7.The Panbio™ COVID-19 Antigen Self-Test is a lateral flow test that detects the nucleocapsid Panbio™ COVID-19 Antigen Self-Test is not intended to detect from defective (non-infectious) virus during the later stages of viral shedding that might be detected by PCR molecular tests.
HSE COVID-19 antigen test validation report June 2021
www.hse.ieA total of seven antigen test have been validated/verified, 6 lateral flow tests (LFT [s) and one microfluidic device with reader. The key findings from this validation work are as follows: • Reverse transcription polymerase chain reaction (RT-PCR) is the gold standard test for detection of SARS-CoV-2. It is the most sensitive technology for ...
CareStart COVID-19 Antigen test - Instructions for Use
www.henryschein.comApr 12, 2021 · The CareStart™ COVID-19 Antigen test is a lateral flow immunochromatographic assay for the detection of extracted nucleocapsid protein antigens specific to SARS-CoV-2 in nasopharyngeal or anterior nasal swab specimens directly collected from …
Center for Clinical Standards and Quality/Survey ...
www.cms.govAug 26, 2020 · CoV-2 or to diagnose a possible case of COVID-19 (e.g., molecular, antigen, antibody) are required to report, regardless of the type of laboratory (type of CLIA certificate) performing the testing. All negative and positive SARS-CoV-2 results must be reported irrespective of the method (e.g., molecular, lateral flow) used.