Search results with tag "Antigen test"
Listen der SARS-CoV-2-Schnelltests zur Fachanwendung und ...
www.bag.admin.chMEDsan GmbH, MEDsan SARS-CoV-2 Antigen Rapid Test 6 1180 Merlin Biomedical (Xiamen) Co., Ltd., SARS-CoV-2 Antigen Rapid Test Cassette 0 2029 MEXACARE GmbH, MEXACARE COVID-19 Antigen Rapid Test 0 1775 möLab, mö-screen Corona Antigen Test 0 1190 MP Biomedicals, Rapid SARS-CoV-2 Antigen Test Card 15 1481
Rapid SARS-COV-2 Antigen Test Card Self-test
www.tga.gov.auRapid SARS-CoV-2 Antigen Test Card is a one step lateral flow test tor the detection of SARS-CoV-2 virus antigen in nasal swabs from individuals suspected of having COVID-19. The Rapid SARS-CoV-2 Antigen Test Card cannot be used as the sole basis to diagnose or exclude SARS-CoV-2 infection. Children under 14 years of age should be assisted by an
INSTRUCTION COVID-19 RAPID GUIDE ANTIGEN TEST …
www.hsa.gov.sgINDICAID™ COVID-19 Rapid Antigen Test is an in vitro diagnostic test for determining the presence of SARS-CoV-2 antigen in direct nasal swab samples or nasopharyngeal swab sample. This test is intended for self-testing and/or professional use. PRINCIPLE During COVID-19 infection, the virus SARS-CoV-2 is found in the upper respiratory tract.
Consent and Registration Form for Rapid COVID-19 Antigen …
www.mhsaa.com5. I understand that my antigen test result will be available in 15-30 minutes. If the result is positive, it will need to be confirmed with a PCR test. 6. I understand and acknowledge that a positive antigen test result is an indication that I need to self-isolate to avoid infecting others until I obtain a negative PCR test result. 7.
Your step-by-step guide to the MP Biomedicals Rapid SARS ...
assets.publishing.service.gov.ukReport all your test results to the NHS Read this whole guide carefully before you start the test. This test may be different to the one you have used before. These instructions for use relate to the MP Biomedicals Rapid SARS-CoV-2 Antigen Test Card, 07AG6007BS only. This guide explains how to test yourself or another person for COVID-19, and ...
HSE COVID-19 antigen test validation report June 2021
www.hse.ieA total of seven antigen test have been validated/verified, 6 lateral flow tests (LFT [s) and one microfluidic device with reader. The key findings from this validation work are as follows: • Reverse transcription polymerase chain reaction (RT-PCR) is the gold standard test for detection of SARS-CoV-2. It is the most sensitive technology for ...
Evaluation of Abbott BinaxNOW Rapid Antigen Test for …
www.cdc.govWhen the pretest probability for receiving positive test results for SARS-CoV-2 is elevated (e.g., in symptomatic persons or in persons with a known COVID-19 exposure), a negative antigen test result should be confirmed by NAAT (1). Despite a lower sensitivity to …
CareStart COVID-19 Antigen test - Instructions for Use
www.henryschein.comApr 12, 2021 · The CareStart™ COVID-19 Antigen test is a lateral flow immunochromatographic assay for the detection of extracted nucleocapsid protein antigens specific to SARS-CoV-2 in nasopharyngeal or anterior nasal swab specimens directly collected from …
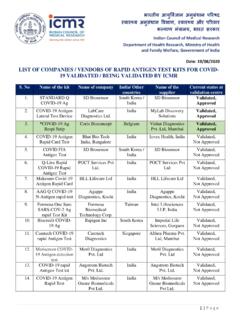
LIST OF COMPANIES / VENDORS OF RAPID ANTIGEN TEST …
stopcorona.tn.gov.inAntigen Lateral test Device (Lab Care Diagnostics Ltd) ... to be added to the well of the lateral flow strip. Read results in 15 mins or earlier after insertion of strip into tube containing sample and dilution buffer. The strip should be discarded after 15 minutes.