Search results with tag "Asrs"
Coronavirus SARS-COV-2: Kurzüberblick Sonderregelungen
www.kbv.deben die Möglichkeit, bei der Behandlung von Opioidabhängigen von den Vorgaben der Betäubungsmittel-Verschreibungsverordnung abzuweichen. Sie dürfen jetzt beispielsweise mehr Patienten behandeln als bisher und können Substitutionsmittel in einer Menge verschreiben, die für bis zu sieben aufeinanderfolgende Tage benötigt wird.
eFILING USER GUIDE FOR VALUE-ADDED TAX
vatcalculator.co.za3 | eFILING USER GUIDE FOR VALUE-ADDED TAX 1.INTRODUCTION Since 2007, the South African Revenue Service (SARS) has been modernising and simplifying tax processes in line with international best practice.
GEN-PEN-05-G01 - South African Revenue Service
www.sars.gov.zaeFiling; At a local SARS branch, by making an appointment on the SARS website. • SARS can either ALLOW, PARTIALLY ALLOW or DISALLOW the request for remission, objection or appeal against PIT and/or CIT admin penalties. The outcome will be communicated through a dispute outcome letter, which will be viewable on eFiling if the RFR1/NOO1/NOA1 ...
GEN-DC-20-G03 - Deferral of Payment Arrangements on ...
www.sars.gov.zaLog on to the SARS eFiling website and navigate to on eFiling where there is outstanding debt against an assessed return in order to initiate the payment arrangement request. Payment arrangement request can be initiated from any of the following places where there is outstanding debt and only one tax type request at a time will be allowed:
GEN-PEN-05-G02 - South African Revenue Service
www.sars.gov.zaThe SARS online query system on the SARS website; and At a local SARS branch, by making an appointment via the SARS website. • The dispute process can be triggered from the following functionalities on eFiling: ... Guide For Provisional Tax External Guide for more – ...
Anti-SARS-CoV-2 Neutralizing Antibodies
www.cdc.govAug 28, 2020 · • In the final model, older age, male sex, persons with Medicare or unknown insurance, and persons with ... May 31, 2020. Cox proportional hazards regression was used to evaluate associations between patient characteristics and time to all …
SARS - CoV - 2 (COVID19) Fact Sheet-
www.cdc.govSARS - CoV - 2 (COVID19) Fact Sheet- There are regulatory considerations that must guide the use of POC instruments for SARS-CoV-2 diagnostic purposes. Testing sites operating a POC diagnostic instrument must have a current certificate via the Clinical Laboratory Improvement Amendments of 1988 (CLIA). During the COVID-19 public
Listen der SARS-CoV-2-Schnelltests zur Fachanwendung und ...
www.bag.admin.chMEDsan GmbH, MEDsan SARS-CoV-2 Antigen Rapid Test 6 1180 Merlin Biomedical (Xiamen) Co., Ltd., SARS-CoV-2 Antigen Rapid Test Cassette 0 2029 MEXACARE GmbH, MEXACARE COVID-19 Antigen Rapid Test 0 1775 möLab, mö-screen Corona Antigen Test 0 1190 MP Biomedicals, Rapid SARS-CoV-2 Antigen Test Card 15 1481
How to Do a SARS-CoV-2 Antigen Rapid Diagnostic Test
terrance.who.intmeans SARS -2 is NOT DETECTED Invalid NO LINE in “C” and a line or no line in “T” means the test is INVALID. Repeat the test using a new (unopened) SARS-CoV-2 Antigen test device and a new sample Produced by the Foundation for Innovative New Diagnostics (FIND) from COVID-19 Antigen RDT manufactures’ Instructions for Use.
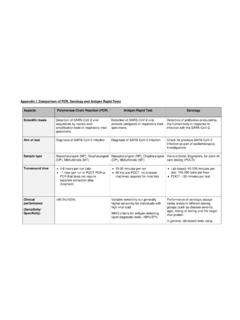
Appendix I: Comparison of PCR, Serology and Antigen Rapid ...
www.moh.gov.sgAppendix I: Comparison of PCR, Serology and Antigen Rapid Tests Aspects Polymerase Chain Reaction (PCR) Antigen Rapid Test Serology Scientific basis Detection of SARS-CoV-2 viral sequences by nucleic acid amplification tests in respiratory tract specimens. Detection of SARS-CoV-2 viral proteins (antigens) in respiratory tract specimens.
2021 HEALTH ADVISORY #39 COVID-19 ORAL ANTIVIRAL ...
www1.nyc.govonly one monoclonal antibody product that is effective for treatment of infection caused by the omicron variant. While supplies remain low, adhere to the NYS DOH guidance on prioritization of anti-SARS-CoV-2 therapies for treatment and prevention of severe COVID-19 and prioritize
Evaluation of Abbott BinaxNOW Rapid Antigen Test for …
www.cdc.govWhen the pretest probability for receiving positive test results for SARS-CoV-2 is elevated (e.g., in symptomatic persons or in persons with a known COVID-19 exposure), a negative antigen test result should be confirmed by NAAT (1). Despite a lower sensitivity to …
Anti-SARS-CoV-2 QuantiVac ELISA (IgG)
www.coronavirus-diagnostics.comAcute EBV infection & heterophilic antibodies 22 100 % Rheumatoid factors 40 100 % Total 1458 99.8 % Borderline results (n = 5 ) were not included. Serial dilutions of the “First WHO International Standard for anti-SARS-CoV-2 immunoglobulin” (NIBSC code: 20/136) were investigated with the Anti-SARS-CoV-2 QuantiVac ELISA (IgG).
FACT SHEET COVID-19 disease (SARS-CoV-2 virus)
covid-19.sciensano.be97% of PCR tests in England which test negative on the S-gene target and positive on other targets were due to the 501Y.V1 variant. S gene drop-out has therefore been used as a proxy for 501Y.V1 (21). Analysis of data in the UK, as of January …
Updated CLIA SARS-CoV-2 Molecular and Antigen Point of ...
www.cms.govUpdated CLIA SARS-CoV-2 Molecular and Antigen Point of Care Test Enforcement Discretion . What is CMS’s policy regarding laboratories performing SARS-CoV-2 molecular and antigen tests authorized by the Food and Drug Administration (FDA) under an Emergency Use Authorization (EUA) for use at the point of care (POC) or in patient care settings
SNPsig SARS-CoV-2 (EscapePLEX) - genesig.com
genesig.comSNPsig® kit Handbook Path-SNPsig®-SARS-CoV-2-EscapePLEX-STED Issue 1.00 Published Date: 3rd June 2021 4 Introduction The novel coronavirus disease 2019 (COVID-19) pandemic, caused by SARS-CoV-2 virus, represents a major threat to health. SARS-CoV-2 has resulted in widespread morbidity and mortality.
Financial Crimes Enforcement Network
www.fincen.govSARs Filed by Money Services Businesses (MSBs)..... 21 SARs Filed by Broker-Dealers ... Use the Narrative Section to indicate that multiple offices of a single financial institution were involved in the suspicious activity and provide the addresses of those locations.
Overview of ICH E2F – Development SafetyDevelopment …
database.ich.orgSubject US IND Annual Report EU Annual Safety Report Short-term trials ≤1year from end of td ≤ 90 days from end of study ti ltrial Regulator feedback On request Not specified Adverse events included All SAEs All SARs Expectedness IB or package insert IB or Summary of Product Characteristics 9
Fact Sheet for Patients Interpreting SARS-CoV-2 RNA ...
www.sonoraquest.comMar 09, 2020 · Fact Sheet for Patients Interpreting SARS-CoV-2 RNA, Qualitative Real-Time RT-PCR Test Results Updated: March 9, 2020 You are being given this Fact Sheet because your sample(s) were tested for the Coronavirus Disease 2019 (COVID9) -1 using the Quest Diagnostic SARS-CoV-2 RNA, Qualitative Real-Time RT-PCR test (“Quest test”) available through
GEN-GEN-51-G01 - SARS Online Query System - External …
www.sars.gov.zaquery; this query must be used to report the taxpayer as an estate to SARS. 14-12-2020 2 Updated the guide with Request to be updated as the Registered Representative query. 31-03-2021 3 Updated the guide with the Request your Tax Compliance Status (TCS) query. 14-06-2021 4 Update the guide with a new query TCS Verification.
Prioritization of Anti-SARS-CoV-2 Monoclonal Antibodies ...
coronavirus.health.ny.govDec 29, 2021 · Prioritization of Anti-SARS-CoV-2 Monoclonal Antibodies and Oral Antivirals for the ... In addition, the most efficacious products should be ... vaccine should be considered for pre-exposure prophylaxis with a long-acting monoclonal antibody (Evusheld).
SAICA GUIDANCE REMOVING AND ADDING TAXPAYER …
saicawebprstorage.blob.core.windows.netSARS can send the OTP and click on <Send OTP> Insert the OTP sent to your email or cell number and click on <Submit>. c) You can view the details of the party requesting the tax type transfer and print the Power of Attorney Click on <Accept> or <Reject>. After you have authorised or rejected the request, click on
Indian SARS-CoV-2 GENOMICS consortium
www.mohfw.gov.inOver the last few weeks, the United Kingdom (UK) has faced a rapid increase in COVID-19 cases in South East England, leading to enhanced epidemiological and virological investigations. Analysis of viral genome sequence data identified that a large proportion of cases belonged to a new single phylogenetic cluster.
City of Joburg Property Company (SOC) Ltd Department of ...
jhbproperty.co.za(pin) issued by sars to enable the organ of state to view the taxpayer’s profile and tax status. 2.3 application for the tax compliance status (tcs) certificate or pin may also be made via e-filing. in order to use this provision, taxpayers will need to register with sars as e-filers through the website www.sars.gov.za.
WHO Emergency Use Listing for In vitro diagnostics (IVDs ...
extranet.who.intNovel Coronavirus (SARS-CoV-2) Real Time Multiplex RT-PCR Kit RR-0485-02 RoW EUL-0486-139-00 50 T/kit 21 May 2020 Fast Track Diagnostics Luxembourg S.à r.l. FTD SARS-CoV-2 11416300 11416284 CE-mark EUL-0502-193-00 32 T/kit 96 T/kit 19 May 2020 Beijing Applied Biological Technologies Co. Ltd., (XABT) Multiple Real-Time PCR Kit for Detection of ...
WHO Emergency Use Listing for In vitro diagnostics (IVDs ...
extranet.who.int30 November 2020 EUL-0497-190-00 Bio-Speedy SARS-CoV-2 (2019-nCoV) qPCR Detection Kit. CE-mark BS-SY-WCOR-304-100 ... Antibody Detection Tests Date Listed EUL Number Product name Regulatory version Product code(s) Manufacturer Packaging 07 December 2020 EUL 0542-118-00 Elecsys Anti-SARS-CoV-2 Qualitative assay for use on the cobas e …
Innate Immune Suppression by SARS-CoV-2 mRNA …
d197for5662m48.cloudfront.netInnate Immune Suppression by SARS-CoV-2 mRNA Vaccinations: The role of G-quadruplexes, exosomes and ... of virus or their protein products, referred to as “subunit vaccines,” has been more technically challenging [1]. In any event, an implicit assumption behind the deployment of any vaccination campaign is that the ... anti-viral as well as ...
Acyclovir for SARS-CoV-2: An old drug with a new purpose
openaccessjournals.comhyperlipidemia, hypothyroidism, osteoporosis, and acid reflux, was initially presented to an urgent care center on October 23, 2020 for symptoms of fever, chills, urine frequency, and fatigue for the past 3 days. The patient underwent nasal swab to test for COVID-19, which resulted NEGATIVE, so she was treated with
Role of Ventilation in Controlling SARS-CoV-2 …
assets.publishing.service.gov.ukcold stores) may pose an enhanced risk (medium confidence). Providing the ventilation rate remains the same, increasing the occupancy of a space increases the probability of airborne transmission by four fold. Exposure risk may be further increased if distances between people are reduced to <2m. (medium confidence).
Summary of Evidence and Information: SARS-CoV-2
optn.transplant.hrsa.govJan 21, 2022 · Summary of Current Evidence and Information– Donor SARS-CoV-2 Testing & Organ Recovery from Donors with a History of COVID-19 . Aim . This document is a summary of evidence and information regarding donor screening for SARS-CoV-2 and considerations for organ acceptance from donors with a history of COVID-19. It is based on peer-reviewed ...
Interim analysis of COVID-19 vaccine effectiveness against ...
www.ecdc.europa.euIn late 2019, a novel severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), which causes coronavirus disease 2019 (COVID-19), emerged. On 11 March 2020, the World Health Organization declared COVID-19 a pandemic. As of 23 October 2021, over 39 million cases and almost 5 million deaths were reported in the EU/EEA [7].
Federal Response to COVID-19: Therapeutics Clinical ...
www.phe.gov1 literature (Dec 22, 2021 Early Remdesivir to Prevent Progression to Severe Covid-19 in Outpatients; DOI: 10.1056/NEJMoa2116846) 2. COVID-19 convalescent plasma with high titers of anti-SARS-CoV-2 antibodies is authorized for the treatment of COVID-19 in patients COVID-19 Convalescent Plasma EUA) Consider
CPT Category I and Proprietary Laboratory Analyses (PLA ...
www.ama-assn.org• Further updates to this document will only occur when there are additional modifications to severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) (coronavirus disease [COVID-19]) codes other than vaccines. • Deleted codes in this document appear with a strikethrough.
How Did the COVID-19 Vaccine Get Developed So Quickly?
portal.ct.gov• Severe acute respiratory syndrome (SARS) and Middle East respiratory syndrome (MERS) are two diseases caused by coronaviruses closely related to the virus that causes COVID-19. Researchers began working on developing vaccines for these diseases after they were discovered in 2003 and 2012, respectively. • None of the SARS vaccines ever made
Gestió de la infecció pel coronavirus SARS-CoV-2 en l ...
canalsalut.gencat.catGestió de la infecció pel coronavirus SARS-CoV-2 en l’àmbit residencial 6 viuen requerirà d’adaptacions en la seva aplicació, respectant sempre les
SARS-CoV-2 Diagnostic Testing - Department of Health
coronavirus.health.ny.govDiagnostic tests detect SARS-CoV-2 in samples collected from your nose or throat. A diagnostic test is the only way to know if you are infected with SARS-CoV-2. These tests can be performed for those with or without symptoms. How does the SARS-CoV-2 diagnostic test work?
CPT Assistant guide: Coronavirus (SARS-CoV-2); September …
www.ama-assn.orgsyndrome coronavirus 2 (SARS-CoV-2). New code 99072 was established to report additional practice expenses incurred during a Public Health Emergency (PHE), including supplies and additional clinical staff time. This article will provide an overview of these new codes. Immunology # 86413 Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2)
EXPEDIENTE DE VACUNACIÓN CONTRA EL VIRUS SARS …
www.huixquilucan.gob.mxEXPEDIENTE DE VACUNACIÓN CONTRA EL VIRUS SARS-COV2 Fecha de la vacunación Folio mivacuna (opcional): D D M M A A NOMBRE(S) APELLIDO 1 APELLIDO 2 Folio captura (opcional) CURP (Clave única de registro de población) Teléfono ó celular 1 Correo Electrónico Teléfono ó celular 1 Calle N. Exterior N. Interior C.P. Municipio Estado
Sotrovimab Product Monograph
ca.gsk.com(Anti-SARS-CoV-2 spike protein monoclonal antibody) HEALTH CANADA HAS AUTHORIZED THE SALE OF THIS COVID-19 DRUG BASED ON LIMITED CLINICAL TESTING IN HUMANS AND/OR QUALITY INFORMATION Sotrovimabis indicated forthe treatment of mild to moderate coronavirus disease 2019 (COVID-19),
Rapid SARS-COV-2 Antigen Test Card Self-test
www.tga.gov.auThe Rapid SARS-CoV-2 Antigen Test Card cannot be used as the sole basis to diagnose or exclude SARS-CoV-2 infection. Children under 14 years of age should be assisted by an adult. SUMMARY COVID-19 is an acute respiratory infectious disease caused by the SARS-CoV-2 virus. Infected people either with or without symptoms can be a source of infection.
CPT Assistant guide: Coronavirus (SARS-CoV-2); August 2020
www.ama-assn.orgSevere Acute Respiratory Syndrome Coronavirus (SARS-CoV-2) Laboratory Testing There is precedence for the the American Medical . Association (AMA) Current Procedural Terminology (CPT ®) Editorial Panel immediately releasing CPT codes when an emergent issue arises. Due to the coronavirus disease (COVID-19) pandemic, this
Similar queries
Coronavirus SARS-COV-2: Kurzüberblick Sonderregelungen, Patienten, EFiling, South African Revenue Service, SARS, SARS eFiling, SARS online query system, External, SARS-CoV-2 Neutralizing Antibodies, Model, Cox proportional hazards regression, 2 Antigen Rapid, Antigen Rapid, Antigen, Rapid SARS, 2 Antigen, Test, 2 Antigen test, Antibody, Anti, Rapid Antigen Test, Antigen test, Anti-SARS-CoV-2 QuantiVac, Heterophilic, FACT SHEET COVID-19 disease SARS-CoV, Financial Crimes Enforcement Network, Multiple, Report, Fact Sheet, Patients, SARS Online Query System - External, Taxpayer, Request, Products, Power of Attorney, Indian SARS-CoV-2 GENOMICS consortium, COVID, Analysis, Acyclovir for SARS-CoV-2, Drug with a new purpose, Hypothyroidism, Symptoms, Role of Ventilation in Controlling SARS-CoV, Risk, Fold, Summary, Evidence, SARS-CoV-2, Severe Acute Respiratory Syndrome, Severe acute respiratory syndrome SARS, Respiratory syndrome, De la infecció pel coronavirus SARS-CoV, SARS-CoV-2 diagnostic, Diagnostic, CPT Assistant, EXPEDIENTE DE VACUNACIÓN CONTRA EL VIRUS SARS