Search results with tag "Sterile"
MDR Labelling Requirements - Medical Device Regulation
www.medical-device-regulation.eu•Sterile package requirements (23.3) • AIMDD already distinguishes sterile pack from trade pack / sales pack; this distinction is new to MDD devices • Mostly a sub-set of existing labelling requirements •“an indication permitting the sterile packaging to be recognized as such,”–i.e. disclaimers, sterile symbol,
Pharmacy - Terminology
philstaver.weebly.comthe chance of contamination. The ability of personnel to manipulate sterile preparations, sterile packaging components and sterile administration devices in such a way to avoid the introduction of viable micro-organisms. Batch Preparation – Compounding of multiple units of the same item’ not for immediate use.
Pharmacy Self-Inspection Worksheet USP 797 – Sterile ...
doh.wa.govsupervise compounding and dispensing of CSPs shall ensure that the following objectives are achieved: 1. Compounding personnel are adequately skilled, educated, instructed, and trained to correctly perform and document the following activities in their sterile compounding duties. a. perform antiseptic hand cleansing and disinfection of nonsterile
Expiration Dates For Sterile Items - UTMB
www.utmb.eduassure that sterile conditions have been maintained. Special attention will be paid to: The outside of the package to make sure that the integrity of the sterile wrap has not been compromised. Medications, antiseptics and other materials inside the pack that have manufacturers’ expiration dates. These will need to be
797 PHARMACEUTICAL COMPOUNDING—STERILE …
www.uspnf.comNov 22, 2019 · sterile.] • Baths and soaks for live organs and tissues • Implants SPECIFIC PRACTICES Repackaging: Repackaging of a sterile product or preparation from its original container into another container must be performed in accordance with the requirements in this chapter. 2 á797ñ Revision Bulletin Official December 1, 2019
Influenza Specimen Collection - Centers for Disease ...
www.cdc.govPlace specimen in sterile viral transport media tube. Note: NP aspirate may not be possible . to conduct in infants. Deep Nasal Swab • Sterile polyester swab (aluminum or plastic shaft preferred) • Viral transport media tube (should contain 1-3 ML of sterile viral transport medium) 1 . Tilt patient’s head back 70 degrees. 2
Pharmacy Self-Inspection Worksheet USP 797 – Sterile ...
doh.wa.govsterile ingredients, products, components, and devices." Click or tap here to enter text. ☐ ☐ ☐ 18 . Compounding involves only transfer, measuring and mixing manipulations using not more than 3 commercially manufactured sterile products and not more than 2 entries into any container. USP Chapter 797 - CSP Microbial Contamination Risk Levels
Cigna Medical Coverage Policy- Therapy Services Acupuncture
static.cigna.comNext Review Date: 4/15/2022 ... 1 Not all outcome measures have MCID’s determined and supported in the literature. Actual significance of these findings ... acupuncture safe when performed by qualified practitioners using sterile needles. The FDA requires that sterile,
GOOD LABORATORY PRACTICES
pharmexcil.comprocess and the conditions under which non-clinical health and environmental safety studies are planned, performed, monitored, ... ðvFor units having both sterile & non sterile products ... involves laboratory skills . Records of training must be kept.
NfG on Maximum shelf-life for sterile products for human use
www.ema.europa.euUNPRESERVED STERILE PRODUCTS General Chemical and physical in-use stability has been demonstrated for x hours/days at y oC. From a microbiological point of view, unless the method of opening/reconstitution/dilution precludes the risk of microbial contamination, the product should be used immediately.
Draft guideline on the sterilisation of the medicinal ...
www.ema.europa.eu89 The guideline concerns only specific requirements relatin g to sterility and sterile products. For other 90 considerations on the manufacturing of the medicinal product, reference is made to other guidance 91 . documents such as Guidelines on Manufacture of the Finished Dosage Form. 92 . 4.1. Manufacturing of sterile medicinal products
Alcohol Textbook 4thEd - University of São Paulo
edisciplinas.usp.brA distillery is never going to be a sterile fermentation and even those of us who have had the good fortune to operate in sterile fermentation conditions know how easily infection can take hold. Since the yeast is a relatively slow-growing microorganism compared to most of the infectious microorganisms (Lactobacillus, etc.),
Cleaning and Disinfection Protocol - Virox Technologies Inc.
info.virox.comSterile water for irrigation is used with the appropriate cleaning agent in any ISO Class 5 area, including work bench or hood surfaces, walls or other surfaces of the area, and any compounding equipment. Sterile water for irrigation should also be used for intermittent hand decontamination
Annex 1 : Manufacture of Sterile Products 2 Document map
www.pda.org5 168 4 Premises 169 170 4.1 The manufacture of sterile products should be carried out in appropriate cleanrooms, entry to 171 which should be through changing rooms that act as airlocks for personnel and airlocks for 172 equipment and materials. Cleanrooms should be maintained to an appropriate cleanliness standard 173 and supplied with air which has passed through filters …
Quality Assurance of Aseptic Preparation Services: Standards
www.rpharms.comLicensed sterile medicinal products are used as ingredients or the ingredients are manufactured sterile in licensed facilities Products will be allocated a shelf life of no more than one week. The shelf life should be supported by stability data All activities should be in accordance with defined NHS guidelines.
AFTERCARE INSTRUCTIONS FOR MOHS/STERILE SURGERY
associatesindermatology.comAFTERCARE INSTRUCTIONS FOR MOHS/STERILE SURGERY o The pressure bandage that was put in place following surgery must remain in place for 48 hours, unless otherwise directed. o The bandage must stay absolutely dry for 48 hours. o If the surgical site is on your face, please sleep with your head elevated on two pillows or sleep in a recliner. o Please avoid sleeping on …
ATTACHMENT B CY 2022 ESRD PPS CONSOLIDATED BILLING …
www.cms.govJan 01, 2022 · administration, laboratory tests, supplies, and services provided for the treatment of ESRD are included in the ESRD PPS. DME ESRD SUPPLY HCPCS FOR ESRD PPS CONSOLIDATED BILLING EDITS HCPCS Code Long Description A4216 STERILE WATER, SALINE AND/OR DEXTROSE, DILUENT/FLUSH, 10 ML A4217 STERILE WATER/SALINE, …
Collection of Urine Midstream - Guidelines
aci.health.nsw.gov.au7. Ask patient to pass first part of urine stream into the toilet/bedpan/bottle 8. Catch midstream specimen in sterile specimen container or sterile kidney dish. Take care not to contaminate the specimen. 9. Allow patient to void remainder of urine. Ensure that the foreskin is reduced over glans 10. Ensure lid is tightened on container.
FOR PHARMACY COMPOUNDING OF NON-STERILE …
www.napra.cacompounding personnel, with a view to guaranteeing the overall quality and safety of non-sterile preparations. The use of other technologies, techniques, materials and procedures may be acceptable, if they have been proven to be equivalent or
Guidance for Addressing Shortages of Sterile Water for ...
nhia.orga Abbreviations: SWFI: Sterile Water for Injection, NS: Sodium Chloride 0.9%, D5W: Dextrose 5% in Water, D5NS: Dextrose 5% in Sodium Chloride 0.9%, LR: Lactated Ringers 1. Neither the FDA nor pharmaceutical manufacturers can make recommendations outside of product labeling. Primary literature or tertiary sources may have
Visio-Classes of Medical Devices - GOV.UK
assets.publishing.service.gov.uk(non-sterile products only) Declaration of conformity Apply CE mark and notified body number Annex V: Production quality assurance. Audit by a notified body including QMS (excluding design) Class IIb medical devices – routes to CE marking Or Annex V1: Inspection quality assurance (non-sterile products only). Audit by a notified body including QMS
chapter 1 Pharmaceutical solutions for oral administration
www.pharmpress.comPharmaceutical solutions are extensively used as dosage forms for the oral administration of therapeutic agents. Pharmaceutical solutions are homogeneous, i.e. the therapeutic agent(s) and excipients are dissolved in the vehicle. Pharmaceutical solutions for oral administration are in non-sterile dosage forms. KeyPoints
2.1 Common Elements for Hospitals (continued)
fgiguidelines.orgb. USP <797>: Pharmaceutical Compounding—Sterile Preparations c. USP <800>: Hazardous Drugs—Handling in Healthcare Settings (1) A room or area for receiving, unpacking, and inventory control of materials used in the pharmacy (2) Work counters and space for automated and manual dispensing activities (3) An extemporaneous compounding area.
ADMINISTRATION RECONSTITUTION For Intramuscular Use ...
www.toronto.caROCEPHIN sterile powder should be stored at a controlled room temperature (between 15-30ºC) and protected from light. Reconstituted Solutions – Stability and Storage Recommendations . 1. For Intramuscular Use: Solutions should be reconstituted immediately before use. If …
Chemistry, Manufacturing, and Controls of Drug Candidates ...
accelerate.ucsf.edudiagrams, sterilization process for sterile products) – Analytical Methods • brief description of test methods and limits (dosage form dependent) – Stability of Drug product • Information to assure the product’s stability during the planned clinical …
Children's Hospital of Eastern Ontario
www.cheo.on.caChildren's Hospital of Eastern Ontario MASTER FORMULA SHEET ... The non-sterile manufacturing formulas are provided for informational purposes for qualified health care professionals. The hospital will not assume responsibility for the continued currency of the information, any errors or omissions, and/or any ...
Glossary EN - updated legal references 08.2004
ec.europa.euGuidelines for the Manufacture of sterile medicinal products. CLEAN/CONTAINED AREA An area constructed and operated in such a manner that will achieve the aims of both a clean area and a contained area at the same time. CONTAINMENT The action of confining a biological agent or other entity within a defined space.
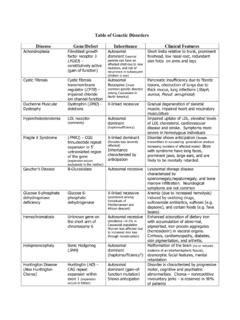
Specimen Collection, Packaging and Transport Guidelines ...
www.mohfw.gov.inSpecimen type Collection Transport to laboratory Storage till testing Comment Nasopharyngeal and oropharyngeal swab Dacron or polyester flocked 4 °C ≤5 days: 4 °C >5 days: -70 °C The nasopharyngeal and oropharyngeal swabs should be placed in the same tube to increase the viral load. Bronchoalveolar lavage sterile container* 4 °C
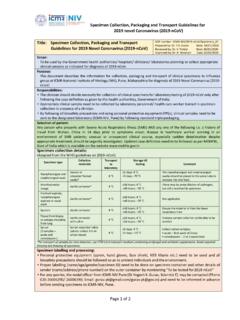
Table of Genetic Disorders - Loyola University Chicago
www.meddean.luc.eduSterile males with long limbs, small genitalia, breast development, and feminine body contours, and learning disabilities Marfan Syndrome Fibrillin-1 gene ... renal cysts, blood in urine, end-stage renal disease and kidney failure. Prader Willi/Angelman (PWS/AS) Deletion of the PWS region and AS gene located at 15q11-q13.
GUIDELINES ON HEATING, VENTILATION AND AIR …
www.who.intWorking document QAS/15.639/Rev.2 page 4 50 BACKGROUND The World Health Organization (WHO)51 published the first edition of the WHO Guidelines on 52 good manufacturing practices for heating, ventilation and air-conditioning systems for non- 53 sterile pharmaceutical dosage forms in WHO Technical Report Series, No. 937, 2006. After …
SUPPLEMENTARY GUIDELINES ON GOOD …
www.who.intSYSTEMS FOR NON-STERILE DOSAGE FORMS Deadline First draft prepared and mailed for comments March 2003 Deadline for receipt of comments 15 April 2003 Collation of comments May 2003 Revision of draft document October 2004 Presentation to Thirty-ninth WHO Expert Committee on Specifications for Pharmaceutical Preparations October 2004
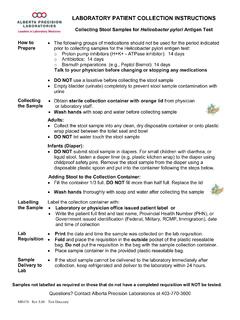
LABORATORY PATIENT COLLECTION INSTRUCTIONS
www.albertahealthservices.cax Obtain sterile collection container with orange lid from physician or laboratory staff. x Wash hands with soap and water before collecting sample Adults: x Collect the stool sample into any clean, dry disposable container or onto plastic wrap placed between the toilet seat and bowl x DO NOT let water touch the stool sample Infants (Diaper):
TEST: Varicella Antibody IgG - Maryland.gov Enterprise ...
health.maryland.govSpecimen: Serum Specimen identification: Label container with patient’s last name, first Name, DOB, specimen type, date and time of collection. Specimen Volume Required: 1-2 ml Specimen Volume Minimum: 1ml Collect: Venipuncture; Red top vacuum tube, transfer serum to sterile tube with leak-proof cap.
leyenda marca de agua - IDENTIFICATION OF POSSIBLE …
www.laquintacolumna.infoAll vials were sealed at the time of processing. Samples were extracted from the respective vials using new sterile micro-syringes and needles. Approximately 10 μl of droplets from each vial were deposited on the microscope slides. In the initial analysis, the samples appear to be flowing in a suspension or hydrogel that
How to Use a Micropipettor-1 - University of California ...
research.mcdb.ucla.edu4. Load a sterile tip. Use blue tips for P1000 pipettes and clear tips for P200 and all smaller sizes. Use filter tips when performing PCR or working with RNA. Close the tip box to maintain sterility. NOTE: Do not allow the pipet tip to touch any object (including your gloves, clothes, hair, skin, bench). 5. Load the sample.
Symbols to be used on labelling (ISO 15223 ... - BSI Group
www.bsigroup.comFeb 25, 2020 · This symbol is only to be used when there is an accompanying Sterile symbol (5.2.1 to 5.2.5 or 5.2.10). This symbol is not to be used on reusable medical devices that are intended to be sterilized between uses. ... To identify the country of manufacture of products. In the application of this symbol, the "CC" shall be replaced by either the two
EU GMP Annex 1 Update 2008 Airborne Particle Counting
www.golighthouse.comAirborne Particle Counting for Pharmaceutical Facilities: Update 2008, EU GMP Annex 1 Lighthouse Worldwide Solutions On February 14th, 2008, The European Commission updated Volume 4 EU Guidelines to Good Manufacturing Practice Medicinal Products for Human and Veterinary Use, Annex 1: Manufacture of Sterile Medicinal Products.
Recommendations for Asepsis for Invasive Surgical ...
albertahealthservices.cathe surgical hand antisepsis. (Figure 2) • Keep hands above the level of the elbow so that w ater flows down during the surgical hand scrub and rinsing process. Avoid contact with the faucet or other potential contaminants. (Figure 3) • Dry hands thoroughly using a sterile towel following a surgical hand scrub. Thorough drying is essential as
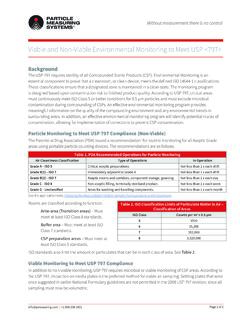
Viable and Non-Viable Environmental Monitoring to Meet …
www.pmeasuring.comThe USP 797 requires sterility of all Compounded Sterile Products (CSP). Environmental Monitoring is an essential component to prove that a cleanroom, or clean device, meets the defined ISO 14644-1 classifications. These classifications ensure that a designated zone is maintained in a clean state. The monitoring program
h55i BIOLOGICAL INDICATORS— RESISTANCE …
www.uspnf.comPlace each carrier in a suitable sterile container containing 100 mL of chilled Purified Water, and sonicate or shake on a reciprocal shaker for an appropriate time. Fifteen minutes or more may be required for ... elastomeric closure or formulated product, its resistance may differ from that determined in Purified Water. That difference may be
ISO 11607 - 10x Medical Device Conference
medicaldeviceevents.comSoCal Institute of Packaging Professionals Chapter, Co-President IoPP Medical Device Packaging Technical Committee, member ... Package Engineering or Product Development must test the product after transit tests ... Eliminates the sample testing requirements of 23°C ±1°C and 50% RH ±2% The sterile barrier system shall allow the product to ...
STERILE TECHNIQUE KEY CONCEPTSAND PRACTICES
www.med.illinois.educoncepts and practices for establishing and maintaining a sterile field. OBJECTIVES After viewing the video and completing the study guide, the participant will be able to: 1. Define sterile technique. 2. Identify the parameters of a sterile field. 3. Describe practices that reduce the spread of infection when preparing or working in a sterile ...
Sterile Water for Irrigation, USP - Baxter
www.baxter.caSterile Water for Irrigation, USP is a sterile, nonpyrogenic solution and contains no bacteriostatic or antimicrobial agents or other substance. Sterile Water for Irrigation, USP is not isotonic and is hemolytic. The composition, osmolarity and approx. pH of Sterile Water for Irrigation, USP is shown in Table 1.
STERILE TECHNIQUE Quick View
info.aorn.orgthe integrity of the filters after removal of the basket by the scrubbed person. - Scrubbed Person: » Lift the inner basket out and above the container without contacting the unsterile surfaces of the table or container. » Before placing instruments on the sterile field, examine the internal chemical indicator for the correct color
Sterile Compounding of Hazardous Drugs
utpharmacyce.learningexpressce.comDefining Hazardous Drugs Hazardous drugs pose potential health risks to workers exposed to them May include:1 Chemotherapy Antiviral drugs Hormone therapy Some bioengineered drugs Miscellaneous National Institute for Occupational Safety and Health (NIOSH) Maintains list of antineoplastics and other hazardous drugs
Similar queries
Medical Device, Sterile package, Sterile, Pharmacy, Preparation, Compounding, Sterile compounding, UTMB, Materials, Container, Specimen, Review, Literature, Process, Skills, Sterile products, Manufacture, Of sterile, Products, University, Sterile water for irrigation, Manufacture of sterile products, Sterile medicinal products, AFTERCARE INSTRUCTIONS FOR MOHS/STERILE SURGERY, Surgery, Services, Collection of Urine Midstream, Urine, PHARMACY COMPOUNDING OF NON-STERILE, Sterile Water, Water, Pharmaceutical, Common Elements for Hospitals continued, Room, Drug, Children's Hospital of Eastern Ontario, Children's Hospital of Eastern Ontario MASTER FORMULA SHEET, Sterile manufacturing, Hospital, Table of Genetic Disorders, Loyola University Chicago, HEATING, VENTILATION AND AIR, World Health Organization, Heating, ventilation and air-conditioning, Sterile pharmaceutical, SUPPLEMENTARY GUIDELINES ON GOOD, NON-STERILE, Processing, Annex, Update 2008 Airborne Particle Counting, Good Manufacturing, Surgical, Surgical hand, Surgical hand scrub, Sterile container, Closure, Chapter, Package, Development, Sterile Water for Irrigation, USP, STERILE TECHNIQUE, After removal, Sterile Compounding of Hazardous Drugs, Hazardous, And other hazardous