Search results with tag "Devices"
Overview of Biomaterials and Their Use in Medical Devices
www.asminternational.orgHandbook of Materials for Medical Devices (#06974G) www.asminternational.org. 2 / Handbook of Materials for Medical Devices Fig. 1 Implant material requirements in orthopedic applications. Source: Ref 2 ... Polyurethanes Blood-contacting devices PVC Tubing PMMA Dental restorations, intraocular lenses, joint replacement (bone cements) Silicones ...
Ensuring critical instruments and devices are appropriate ...
www.jointcommission.orgformulation and scientific validation of reprocessing instructions for reusable medical devices. Manufacturers of medical devices must submit evidence to the FDA to demonstrate that the device to be marketed is safe and effective. Reprocessing instructions for medical devices should be validated. However, because of the greater risks to the ...
Low Cost, Low Power, Differential ADC ... - Analog Devices
www.analog.comlicense is granted by implication or otherwise under any patent or patent rights of Analog Devices. Trademarks and register ed trademarks are the property of their respective owners. One Technology Way, P.O. Box 9106, Norwood, MA 020629106, U.S.A. - Tel: 781.329.4700 www.analog.com Fax: 781.461.3113 ©2004–2012 Analog Devices, Inc.
MDCG 2021-25
ec.europa.euMedical Devices Medical Device Coordination Group Document MDCG 2021-25 Page 1 of 11 MDCG 2021-25 Regulation (EU) 2017/745 - application of MDR requirements to ‘legacy devices’ and to devices placed on the market prior to 26 May 2021 in accordance with Directives 90/385/EEC or 93/42/EEC October 2021
HMC451LP3 / 451LP3E - Analog Devices
www.analog.comfor price, delivery, and to place orders: Analog Devices, inc., one Technology way, p.o. Box 9106, norwood, mA 02062-9106 phone: 781-329-4700 • order online at www.analog.com Application support: phone: 1-800-AnAloG-D Amplifiers - l ine A r & p ower - sm T 2 HMC451LP3 / 451LP3E v02.0121 GaAs PHEMT MMIC MEDIUM POWER AMPLIFIER, 5 - 18 GHz
4-Channel, Low Noise, Low Power, 24-Bit, Sigma-Delta ADC ...
www.analog.comSigma-Delta ADC with PGA and Reference Data Sheet AD7124-4 Rev. D Document Feedback Information furnished by Analog Devices is believed to be accurate and reliable. However, no responsibility is assumed by Analog Devices for its use, nor for any infringements of patents or other rights of third parties that may result from its use.
Special Education EC–12 (161)
www.tx.nesinc.comG. Knows how to make informed decisions about types and levels of assistive technologies, devices and services for students with various needs, collect and analyze information about a student’s environment and curriculum to identify and monitor assistive technology needs and support the use of assistive technologies, devices and services.
The Manual Cleaning Process - IAHCSMM
myhspa.orgcombined with practical skills, ensures that reusable medical devices are safe for employee safety 3. Review the manual cleaning process 4. Address the need for quality assurance ... organic material left on the devices after cleaning lowers the effectiveness of the disinfection or sterilization process, and may lead to subsequent infection. It ...
25 MHz to 3000 MHz Fractional-N PLL with Integrated VCO ...
www.analog.comFractional-N PLL with Integrated VCO Data Sheet HMC832A Rev. B Document Feedback Information furnished by Analog Devices is believed to be accurate and reliable. However, no responsibility is assumed by Analog Devices for its use, nor for any infringements of patents or other rights of third parties that may result f rom its use.
Impact of changes under the new EU Medical Devices …
www.medtecheurope.orgMay 26, 2020 · has been introduced under the regulation (MDR, Article 120). The MDR provides a gradual transition into the new regulatory framework to avoid market disruption and allow a smooth transition from the directives to the regulation. Some medical devices with certificates issued by notified bodies under the directives may
2 A, Ultralow Noise, High PSRR, RF Linear Regulator Data ...
www.analog.comHigh PSRR, RF Linear Regulator Data Sheet ADP7158 Rev. C Document Feedback Information furnished by Analog Devices is believed to be accurate and reliable. However, no responsibility is assumed by Analog Devices for its use, nor for any infringements of patents or other rights of third parties that may result from its use.
16 Channel, 14-Bit, 65 MSPS, Serial LVDS, 1.8 V ADC
www.analog.com16 Channel, 14-Bit, 65 MSPS, Serial LVDS, 1.8 V ADC Data Sheet AD9249 Rev. 0 Document Feedback Information furnished by Analog Devices is believed to be accurate and reliable.
FEATURES DESCRIPTIO U - Analog Devices
www.analog.comCMRR Common Mode Rejection Ratio VCM = 0.1V to 3.2V 90 103 87 102 dB PSRR Power Supply Rejection Ratio VS = ±2V to ±18V, VO = 0V 100 117 97 116 dB Maximum Output Voltage Swing Output Low, 600Ω to GND 615 6 18 mV Output High, 600Ω to GND 3.2 3.8 3.1 3.8 V IS Supply Current 380 630 400 680 µA
AN-686 APPLICATION NOTE - Analog Devices
www.analog.comI2C state machine will determine the effectiveness of the clocking approach.) The method is quite simple. It is the master’s job to recover the bus and restore control to the main program. When the master detects the SDA line stuck in the low state, it merely needs to send some additional clocks and gener-ate a STOP condition.
锁相环常见问题解答 - Analog Devices
www.analog.com锁相环常见问题解答 亚洲技术支持中心 电话:4006 100 006 Email:china.support@analog.com 5 第1章 简介 1.1 ADI 公司锁相环产品概述 ADI 是高性能模拟器件供应商,在锁相环领域已有十多年的的设计经验。
215MHz, Rail-to-Rail Output, 1.1nV/ - Analog Devices
www.analog.com1 623012fc LT6230/LT6230-10 LT6231/LT6232 Typical applicaTion FeaTures DescripTion 215MHz, Rail-to-Rail Output, 1.1nV/√Hz, 3.5mA Op Amp Family The LT®6230/LT6231/LT6232 are single/dual/quad low noise, rail-to-rail output unity-gain stable op amps that
ADXL103/ADXL203 (Rev. F) - Analog Devices
www.analog.comHigh performance, single-/dual-axis accelerometer on a single IC chip 5 mm × 5 mm × 2 mm LCC package 1 mg resolution at 60 Hz Low power: 700 μA at V S = 5 V (typical) High zero g bias stability High sensitivity accuracy −40°C to +125°C temperature range X and Y axes aligned to within 0.1° (typical) Bandwidth adjustment with a single ...
VON DUPRIN Installation Instructions 98/9975 Series ...
www.thebuilderssupply.comInstallation Instructions ® 98/9975 Series Mortise Exit Device This product is covered by the following patent numbers: 3,767,238 4,427,223 3,854,763 4,466,643 4,167,280 4,741,563 Devices covered by these instructions: 98/9975 Mortise Exit Device 98/9975-F (Fire) Mortise Exit Device CD98/9975 (Cylinder Dogging) Mortise Exit Device
CN-0385 - Analog Devices
www.analog.com(ADC) 驱动器,以及一个用于采样有效通道信号的18 位、 2.0 MSPS精密PulSAR® ADC。提供0.4、0.8、1.6 和3.2 增 益配置。 turbo模式下,系统最大采样速率为2 MSPS;正常模式下为 1.5 MSPS。通道切换逻辑与ADC 转换同步,最大通道切换 速率为1.5 MHz。
Evaluating the HMC7044 Dual Loop Clock Jitter Cleaner
www.analog.comevaluation software is C:\Program Files (x86)\Analog Devices\Jitter Attenuator\. 3. After the installation completes, the user is prompted to install the drivers for the USB interface. STARTING THE SOFTWARE After the evaluation software is installed, run the software as follows: 1. Power up and connect the evaluation board to the PC. 2.
Opto-electronic protective devices | 8020197 - SICK
cdn.sick.com• Safe integration into different control Saves time during configuration and diagnostics thanks to user-friendly Safety Designer software • Saves time during commissioning and The highest level of flexibility when adjusting the vehicle speed and direction • Designer software, multi-color display and Flexible connection to different control
EK1HMC7043LP7F User Guide - Analog Devices
www.analog.com1. Apply 0 V to VCCIN (TP14). 2. Apply 3.3 V to 3.3V_MAIN (TP15). 3. Remove R245 and FB20. 4. Populate R12 (to isolate the serial peripheral interface (SPI) board current consumption from the HMC7043). Connect a bench 3.3 V power supply to each of the supply pins on the 3.3 V main headers (TP15 and TP16).
MANUAL ON BORDERLINE AND CLASSIFICATION IN THE …
ec.europa.eu1 . manual on borderline and classification in the community regulatory framework for medical devices. version . 1.22 (05-2019) p. lease note: t. he views expressed in this manual are not legally binding; only the . e. uropean . c. ourt of . j. ustice (“c. ourt
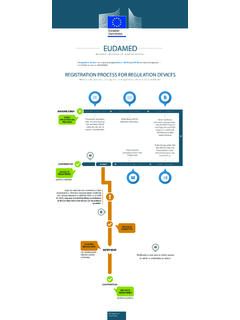
EUDAMED - ec.europa.eu
ec.europa.euDEVICE IS REGISTERED SUBMIT START REGISTRATION PROCESS DEVICE IS REGISTERED DEVICE IS SUBMITTED CONFIRM DEVICE DATA ˜˚˛˝˙˚ˆˇ˝˘ ˘ ˛ ˇ ˙ REGISTRATION PROCESS FOR REGULATION DEVICES A Regulation Device has to have an assigned Basic UDI-DI and UDI-DI and has to be registered in the ‘UDI/Device module’ of …
IP VIDEO DOOR STATION - DoorBird
www.doorbird.comD1101V SURFACE-MOUNT IP VIDEO DOOR STATION Compact Edition 1 Call Button Image sensor technology. ... for mobile devices via WiFi, 3G, 4G, 5G API Microphone • Clear voice transmission. ... Surface-mounted, flush-mounted version sold …
Reference Guide MFC-J5330DW MFC-J2330DW - Brother
download.brother.comfor Brother iPrint&Scan ... mobile devices and computers. 1 General Information 5. 8. [Stored Faxes:] 8 ... 1 From an application, such as Apple TextEdit, click the File menu, and then select Print. 2 Select Brother MFC-XXXX (where XXXX is the name of your model). 3 Click Print.
LT4356-3 (Rev. D) - Analog Devices
www.analog.comBack-to-back FETs can be used in lieu of a Schottky diode for reverse input protection, reducing voltage drop and power loss. The SHDN input turns off the part, including the auxiliary amplifier, and reduces the quiescent current to less than 7µA. n Automotive/Avionic Surge Protection n Hot Swap/Live Insertion
KNOW YOUR LAB EQUIPMENTS - Sri Ramkrishna Sarada …
www.srsvidyamahapitha.orgnear the top that indicates the level at which the volume of the liquid is equal to the volume written on the outside of the flask. These devices are often used when solutions containing dissolved solids of known concentration are needed. Graduated cylinder Graduated cylinders are used to transfer liquids with a moderate degree of accuracy.
Single-/Dual-Supply, High Voltage Isolated IGBT Gate ...
www.analog.comIGBT Gate Driver with Miller Clamp Data Sheet ADuM4135 Rev. D Document Feedback Information furnished by Analog Devices is believed to be accurate and reliable. However, no ... 5 kV ac for 1 minute per UL 1577 . CSA Component Acceptance Notice 5A . DIN V VDE V 0884-10 (VDE V 0884-10):2006-12 . V IORM = 849 V peak (reinforced/basic)
Transimpedance Amplifiers for Wide Range ... - Analog Devices
www.analog.comThe LTC6268 offers 500MHz gain bandwidth, enabling the single-stage circuits shown in the LTC6268 data sheet from 20kΩ transimpedance gain with 65MHz bandwidth to 499kΩ transimpedance gain with 11.2MHz bandwidth. With just 0.45pF input capacitance, the LTC6268 contributes only a small portion of the total circuit capacitance, preserving high
AD9826 Complete 16-Bit Imaging Signal ... - Analog Devices
www.analog.com22 VINB AI Analog Input, Blue Channel 23 CML AO Internal Bias Level Decoupling 24 VING AI Analog Input, Green Channel 25 OFFSET AO Clamp Bias Level Decoupling 26 VINR AI Analog Input, Red Channel TYPE: AI = Analog Input, AO = Analog Output, DI = Digital Input, DO = Digital Output, P = Power. PIN CONFIGURATION TOP VIEW (Not to Scale) 28 27 26 25 ...
AP English Literature and Composition - College Board
secure-media.collegeboard.orgcontrol over the elements of composition in language appropriate to the discussion of prose. These focused ... Zenobia through the use of literary techniques. The introduction identifies the literary devices to be analyzed (“first person perspective, lavish imagery, and probing dialogue ”) and notes the complexity of the narrator’s ...
Driver Support for Atmel CryptoAuthentication Devices ...
ww1.microchip.comname suggests, the boiler-plate is effectively a template. 2. The second part is the dynamic data that changes from device to device. Dynamic data is used to drop into the certificate to make it a valid X.509 certificate. The Certificate API helps store the dynamic data of a certificate into the ATECC device. When the full X.509
LT3120 (Rev. 0) - Analog Devices
www.analog.comMonolithic Buck-Boost Converter The LT®3120 is a high efficiency 26V monolithic buck-boost converter. Extensive feature integration and very low resistance internal power switches minimize the total solution footprint for even the most demanding applica-tions. A proprietary 4-switch PWM architecture provides
LTC3108 (Rev. D) - Analog Devices
www.analog.comVOUT Quiescent Current VOUT = 3.3V, VOUT2_EN = 0V 0.2 µA VAUX Quiescent Current No Load, All Outputs Charged 6 9 µA LDO Output Voltage 0.5mA Load l 2.134 2.2 2.266 V LDO Load Regulation For 0mA to 2mA Load 0.5 1 % LDO Line Regulation For VAUX from 2.5V to 5V 0.05 0.2 % LDO Dropout Voltage ILDO = 2mA 100 200 mV LDO Current Limit VLDO = 0V l 4 ...
MT-046: Op Amp Settling Time - Analog Devices
www.analog.comsource capable of generating a pulse of sufficient flatness. In other words, if the op amp under test has a settling time of 20 ns to 0.1%, the applied pulse should settle to better than 0.05% in less than 5 ns. PULSE GENERATOR (50ΩOUTPUT) DUT 100Ω 100Ω 100Ω R2 R1 + – +0.5V –1.8V –1V 0V 50ΩCOAX "FLAT" REGION MAKE AS SHORT AS POSSIBLE ...
Two Selectable Inputs, 12 LVPECL Outputs, SiGe Clock ...
www.analog.comthe Analog Devices, Inc., proprietary XFCB3 silicon germa-nium (SiGe) bipolar process. This device is designed for high speed applications requiring low jitter. The device has two selectable differential inputs via the IN_SEL control pin. Both inputs are equipped with center tapped, differential, 100 Ω on-chip termination resistors. The inputs
AN57 - Video Circuit Collection - Analog Devices
www.analog.comneed to have an appropriate offset applied to preserve linear amplifi er operation over the intended signal swing. For systems that lack an available negative supply, the LT1983-3 circuit shown in Figure 5 can be used to easily produce a local-use –3V that can simplify an overall cable-driving solu-
3 MSPS, 12-/10-/8-Bit ADCs in 6-Lead TSOT ... - Analog Devices
www.analog.comlow power, successive approximation analog-to-digital converters (ADCs), respectively. The parts operate from a single 2.35 V to 3.6 V power supply and feature throughput rates of up to 3 MSPS. The parts contain a low noise, wide bandwidth track-and-hold amplifier that can handle input frequencies in excess of 55 MHz.
TOUGHBOOK G2 - Panasonic
na.panasonic.com• Optional dedicated GPS (u-blox NEO M8N) • Dual high-gain antenna pass-through • ®Intel Wi-Fi 6 AX201 • ®Bluetooth v5.1 (Class 1) ... 6 The module firmware and the device antenna system have obtained technical approval by commercial carriers in the US. It is customer’s responsibility to assure that the module firmware and the device
AN127: Flash Programming via the C2 Interface - Silicon Labs
www.silabs.comThe length of the WAIT field is controlled by the target device. During the WAIT field, the target device transmits 0’s on the C2D pin until it has finished writing to the target Data register. To indicate the write complete status, the target device transmits a 1 to terminate the WAIT field. AN127: Flash Programming via the C2 Interface C2 ...
NI 6289 Device Specifications - National Instruments
www.ni.comMaximum output level ±11 V Output coupling DC Output impedance 0.2 Ω Output current drive ±5 mA Overdrive protection ±25 V Overdrive current 20 mA 1 An AO channel cannot be a reference or offset to itself. 2 When the USB Screw Terminal device is powered on, the analog output signal is not defined until after the USB configuration is complete.
BELL WEATHERPROOF BOXES, COVERS AND LIGHTING
www.bell-electrical.comdevice(s) or equipment supported by that yoke or strap. A device or utilization equipment wider than a single 50 mm (2 in.) device box as described in Table 314.16(A) shall have double volume allowances provided for each gang required for mounting. (5) Equipment Grounding Conductor Fill. Where one or more equipment grounding conductors
u-center User guide - U-blox
content.u-blox.com• Check our homepage (https://www.u-blox.com) to ensure that your GNSS device, firmware and the u-center software are the latest versions. • Refer to our web based information service. 1.3.1 Worldwide Web Our website (www.u-blox.com) is a rich pool of information. Product information and technical documents can be accessed 24/7.
Be ready BEFORE the emergency - Centers for Disease ...
www.cdc.govTemperature monitoring device currently stored in refrigerator can be used, as long as there is a device to measure temperatures for any remaining vaccines. Why do you need cardboard, bubble wrap, and conditioned frozen water bottles? ... – Transfer boxes of vaccines quickly to storage refrigerator. Troubleshooting – If there has been a ...
Cyclone V Device Handbook, Volume 1: Device Overview …
www.intel.coman FPGA in a single Cyclone V SoC FPGA—supports over 100 Gbps peak bandwidth with integrated data coherency between the processor and the FPGA. February 2012 CV-51001-1.2. ... Interface peripherals—10/100/1000 Ethernet media access control (MAC), USB 2.0 On-The-GO (OTG) controller, serial peripheral interface (SPI), Quad SPI flash ...
AFFIDAVIT IN SUPPORT OF CRIMINAL COMPLAINTS
bloximages.newyork1.vip.townnews.comcellular telephone, a laptop computer, and a wireless charging device. 7. Task Force Officers traveled to the Best Western Hotel to begin reviewing security footage and SA Eilerman contacted Auglaize County Sheriff's Office Detective Timothy Rammel to initiate a joint investigation into the whereabouts of T.H. 8.
u-blox GNSS Sensor and VCP Device Driver
content.u-blox.comThe u-blox USB Sensor Device Driver connects any u-blox GNSS positioning chips and modules to the Windows Sensor and Location Platform. The u-blox GNSS Sensor Device Driver conforms to Microsoft's Windows Driver Model. It is based on the Windows User Mode Driver Framework (UMDF) and supports the USB suspend mode ...
Microsoft Azure IoT Reference Architecture Version 2.1 9 ...
download.microsoft.comcloud, 2) a cloud gateway service, or hub, to securely accept that data and provide device management capabilities, 3) stream processors that consume that data, integrate with business processes, and place the data into storage, and 4) a user interface to visualize telemetry data and facilitate device management. Following, these subsystems are ...
Similar queries
In Medical Devices, Medical Devices, Devices PVC, Devices, Reusable medical devices, Analog Devices, Medical, Medical Device, REGULATION, HMC451LP3 / 451LP3E, Analog, Sigma, Delta ADC, Data, Cleaning, Fractional-N PLL with Integrated VCO, Linear Regulator Data, Linear Regulator Data Sheet, 65 MSPS, Serial LVDS, Rejection Ratio, PSRR Power Supply Rejection Ratio, Supply, Master, Rail, ApplicaTion, Performance, Installation Instructions 98/9975 Series, Installation Instructions ® 98/9975 Series Mortise, Device, PulSAR, Evaluating the HMC7044 Dual Loop Clock Jitter, Software, Opto-electronic protective devices, Safe, VCCIN, Community regulatory framework for medical devices, Regulation Device, Surface, MOUNT, Brother, For Brother iPrint, Apple, FETs, Volume, Liquid, Graduated cylinder Graduated, Clamp, Circuits, Channel, Digital, Elements, Literary techniques, Literary devices, Driver Support for Atmel CryptoAuthentication Devices, Template, LT3120, Buck, Power, Switch, Quiescent Current, LDO Current, Silicon, Linear, To-digital converters, Blox, Field, Device Specifications, Maximum, Overdrive, BELL WEATHERPROOF BOXES, COVERS AND LIGHTING, Transfer, Volume 1: Device Overview, 1000 Ethernet, Gateway