Search results with tag "Polycystic kidney disease"
Chronic Kidney Disease in the United States, 2021
www.cdc.gov*Includes polycystic kidney disease, among other causes. Learn about CKD from a primary care doctor or a kidney doctor (nephrologist) to better understand treatment options and protect the kidneys. People with glomerulonephritis, polycystic kidney disease, or other kidney disease should talk about specific treatment options with a kidney doctor.
Chronic Kidney Disease in the United States, 2019
www.cdc.govthan 18 years, polycystic kidney disease and glomerulonephritis (inflammation of the kidneys) are the main causes of ESKD. Reported Causes of End-stage Kidney Disease in the United States N=726,331 (all ages, 2016) Source: US Renal Data Sytem *Includes polycystic kidney disease, among other causes. 277382-C March 05, 2019 Early Death
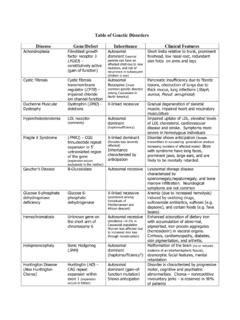
Table of Genetic Disorders - Loyola University Chicago
www.meddean.luc.eduPolycystic Kidney Disease Mutations in either polycystin-1 (PKD1) or polycystin-2 (PKD2) gene Autosomal dominant (disease appears to follow a “two-hit model”, requiring the loss of both alleles of PDK1 or PDK2 for the disease to be evident. Heterozygous individuals are predisposed to polycystic kidney disease because they are likely to
Chronic Kidney Disease (CKD) Algorithm
generalpracticemedicine.org- have a family history of polycystic kidney disease and are aged over 20yrs - have stage 4 or 5 CKD • Advise people with a family history of inherited kidney disease about the implications of an abnormal result before arranging the scan Box 10 - Immunisation • Offer annual influenza vaccination to all patients with confirmed CKD
2 (CKD) and not know it. How can I find out if I have CKD? 3
www.farxiga.comFARXIGA is not for use to improve blood sugar (glucose) control in adults with type 2 diabetes who have moderate to severe kidney problems, because it may not work. FARXIGA is not for people with certain genetic forms of polycystic kidney disease, or who are taking or have recently received immunosuppressive therapy to treat kidney disease.
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS
www.ema.europa.eudominant polycystic kidney disease (ADPKD) in adults with chronic kidney disease (CKD) stage 1 to 4 at initiation of treatment with evidence of rapidly progressing disease (see section 5.1). ... Tolvaptan may cause adverse reactions related to water loss such as …
Farxiga Prescribing Information
den8dhaj6zs0e.cloudfront.netpatients with polycystic kidney disease or patients requiring or with a recent . history of immunosuppressive therapy for the treatment of kidney disease. FARXIGA is not expected to be effective in these populations. (1)----- DOSAGE AND ADMINISTRATION ----- • Assess volume status and correct volume depletion before initiating. ...
Urinalysis: A Comprehensive Review
www.aafp.orgMar 15, 2005 · Medullary sponge kidney ... Polycystic kidney disease Renal artery embolism Renal vein thrombosis Sickle cell disease or trait Tubulointerstitial causes Vascular cause Urologic causes
Diabetes Insipidus - National Institute of Diabetes and ...
www.niddk.nih.goving polycystic kidney disease, sickle cell disease, kidney failure, partial blockage of the ureters, and inherited genetic disorders. Sometimes the cause of nephrogenic DI is never discovered. Desmopressin will not work for this form of DI. Instead, a person with nephrogenic DI may be given hydrochlorothiazide (HCTZ) or indomethacin.
RENAL PHYSIOLOGY AND BODY FLUIDS
downloads.lww.comChronic kidney disease is usually progressive and may lead to renal failure. Common causes include diabetes mellitus, hypertension, inflam-mation of the glomeruli (glomerulonephritis), urinary reflux and infections (pyelonephritis), and polycystic kidney disease. Renal damage may occur over many years and may be unde-
FULL PRESCRIBING INFORMATION - Food and Drug …
www.accessdata.fda.govJYNARQUE is indicated to slow kidney function decline in adults at risk of rapidly progressing autosomal dominant polycystic kidney disease (ADPKD). 2 DOSAGE AND ADMINISTRATION 2.1 Recommended Dosage The initial dosage for JYNARQUE is 60 mg orally per day as 45 mg taken on waking and 15 mg taken 8 hours later.
サムスカ錠 30 mg - pmda.go.jp
www.pmda.go.jp略号一覧 略号 省略していない表現 ADPKD autosomal dominant polycystic kidney disease 常染色体優性多発性嚢胞腎 AUC 血漿中濃度‐時間曲線下面積
Similar queries
Chronic Kidney Disease in the United States, Polycystic kidney disease, Kidney, Kidney disease, Chronic Kidney Disease, Table of Genetic Disorders, Loyola University Chicago, Disease, SUMMARY OF PRODUCT CHARACTERISTICS, Tolvaptan, Prescribing Information, Diabetes Insipidus, FULL PRESCRIBING INFORMATION, Food and Drug