1. The Wittig Reaction - CCC/UPCMLD
Feb 12, 2007 · ratios that depend on the level of stabilization (charge delocalization) of the ylide. The Wittig reaction must involve a mechanism other than the betaine pathway. A synchronous ... An interplay of 1,2- and 1,3-steric interactions decides which diastereomeric oxaphosphetane will be favored. It is important to recall,
Tags:
Information
Domain:
Source:
Link to this page:
Please notify us if you found a problem with this document:
Documents from same domain
Basics of LC/MS (5988-2045EN) - University of …
ccc.chem.pitt.eduContents Why Liquid Chromatography/Mass Spectrometry? 4 Instrumentation 6 Ion Sources 6 Electrospray ionization 7 Atmospheric pressure chemical ionization 8
Reversed-phase HPLC Bu ers
ccc.chem.pitt.edu9 Reversed-phase HPLC Bu!ers High-quality bu!ers (solutions, solids or concentrates) Shyam Verma shyam.verma@sial.com Consideration of …
HPLC Troubleshooting Cover - CCC/UPCMLD
ccc.chem.pitt.edu1 HPLC Troubleshooting Uwe D. Neue, Waters Corporation 1. Column Life-Time 2. Variable Retention Times 3. Drifting Retention Times …
Drying of Organic Solvents: Quantitative …
ccc.chem.pitt.eduDOI: 10.1021/jo101589h J. Org. Chem. XXXX, XXX, 000–000 A rXXXX American Chemical Society pubs.acs.org/joc Drying of Organic Solvents: Quantitative Evaluation of the ...
Evaluation, Quantitative, Organic, Solvents, Drying, Drying of organic solvents, Quantitative evaluation of
NMR Chemical Shifts of Trace Impurities: Common …
ccc.chem.pitt.eduNMR Chemical Shifts of Common Laboratory Solvents as Trace Impurities Hugo E. Gottlieb,* Vadim Kotlyar, and Abraham Nudelman* Department of Chemistry, Bar …
Chemical, Impurities, Common, Trace, Shifts, Solvents, Nmr chemical shifts of trace impurities
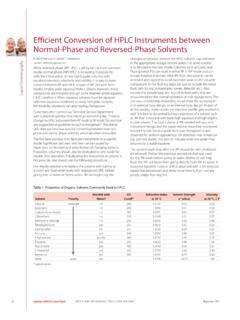
E cient Conversion of HPLC Instruments between Normal ...
ccc.chem.pitt.edu6 Liquid Chromatography sigma-aldrich.com/hplc ORDER: 800-247-6628 (US ONLY) / 814-359-3441 Reporter 28.5 E!cient Conversion of HPLC Instruments between
Between, Intec, Instruments, Conversion, Hplc, Cient conversion of hplc instruments between
Incompatibilities of concentrated nitric acid: Never mix ...
ccc.chem.pitt.eduGroup 11: Olefins (cont.) Propylene tetramer (dodecene) 1-Tetradecene 1-Tridecene Turpentine 1-Undecene Group 12: Petroleum Oils Asphalt Gasolines Casingead Automotive Aviation Jet Fuels JP-1 (kerosene) JP-3 JP-4 JP-5 (kerosene, heavy) Kerosene Mineral spirits Naphtha (non aromatic) Naphtha Solvent Stoddard solvent VM&P Oils Absorption oil
Acid, Olefins, Concentrated, Nitric, Incompatibilities of concentrated nitric acid, Incompatibilities
D.H. Ripin, D.A. Evans pKa's of Inorganic and Oxo-Acids ...
ccc.chem.pitt.eduD.H. Ripin, D.A. Evans 19-20 9 13 11 24.5 H2 ~36 *Values <0 for H2O and DMSO, and values >14 for water and >35 for DMSO were extrapolated using various methods. HCCH Substrate SubstrateSubstrate pKa H2O (DMSO) Substrate AMIDES HYDROCARBONS pKa H2OH(DMSO) pKa pKa2O (DMSO) H2O (DMSO) CH4 CH2=CHCH3 PhH CH2=CH2 PhCH3 …
Related documents
DISPERSANT PRODUCT GUIDE - Uniqchem
www.uniqchem.com1.1.1 Electrostatic stabilization is only working in a water based application. When two particles having the same charges approaching each other will result in a repelling effect. The resulting Coulomb-repulsion of the charged particles allows the system to remain stable. 1.1.2 Steric stabilization suited for water and
Guide, Product, Stabilization, Dispersant, Steric, Steric stabilization, Dispersant product guide
Wetting and Dispersing Additives for Epoxy Applications ...
ebooks.byk.comand/or steric stabilization. Both stabilization measures are described below. Deflocculation generates rather Newtonian flow characteris-tics along with generally reduced viscosity. This, in turn, im-proves leveling behavior and enables higher pigment loading. Due to the small particle size of the deflocculated pigments,
Additives, Stabilization, Wetting, Steric, Dispersing, Steric stabilization, Wetting and dispersing additives
Surfactants – classification, features and applications
www.lcpe.uni-sofia.bgSteric stabilization by nonionic surfactants Nonionic surfactants. Role of surfactant micelles Solution rheology (shampoos, dish-washing gels) higher solution viscosity Structural forces in foam films: Control of micelle shape: mixtures (SLES+CAPB), counterions (Ca2+), temperature (for EO surfactants)
Zeta potential - An introduction in 30 minutes
www.research.colostate.eduSteric stabilization is simple, requiring just the addition of a suitable polymer. However it can be difficult to subsequently flocculate the system if this is required, the polymer can be expensive and in some cases the polymer is undesirable e.g. when a ceramic slip is cast and
Metallic Nanoparticle: A Review - Biomedres.us
biomedres.usSo, this stabilization can be achieved either by steric exclusion or electrostatic stabilization by using a capping agent such as surfactant, polymer, solid support or ligand with suitable functional groups. Electrostatic stabilization
Review, Nanoparticles, Metallic, Stabilization, A review, Steric, Metallic nanoparticle
Reactions of Alkyl Halides - Towson University
tigerweb.towson.edugroup, alleviates both the electronic and steric issues seen in the frontside attack. This is a one-step concerted (i.e. happens all at once) process where all the bond formations and breakages ... Both are extremely good leaving groups due to resonance stabilization: S CH 3 O O SCH 3 O O O O 3. Solvent: The rate of the reaction can be affected ...
IONIC REACTIONS — NUCLEOPHILIC SUBSTITUTION AND ...
www.austincc.eduthe stabilization of the transition state, and thereby increases the free energy of activation, thus decreasing the rate of reaction. 6.18 (Most reactive) (Least reactive) CH 3OSO 2CF 3 > 3I 3Br 3Cl 3F 14 3OH. ... N2 attack because of the steric hindrance. (A very small amount of S
Reactions, Stabilization, Steric, Ionic, Nucleophilic, Ionic reactions nucleophilic
Reaction Mechanisms - IITPK
www.iitpk.comelectron release and stabilization of R +. The effect is opposite in S. N. 2 reactions +because bulky R’s sterically hinder formation of, and raise + ∆ H for, the transition state. (d) Strong nucleophiles favor S N 2 reactions and do not affect S N 1 reactions . Illustration :
What Makes a Good Nucleophile? - University of Minnesota
www1.chem.umn.edubecause of TS stabilization, not product stabilization + + + + What Makes a Good Leaving Group? 2. Polarizable. (Stabilizes transition state.) So, I is a better leaving group than F . I F good overlap, stabilization poor overlap Interesting consequence: I is both a good nucleophile and a good leaving group.