Gemcitabine 200mg and 1 mg Powder for Solution for ...
MHRA PAR; GEMCITABINE 200 MG AND 1 G POWDER FOR SOLUTION FOR INFUSION, PL 20176/0048-9 6 The manufacturing process is referenced to a drug master file (DMF).
Tags:
Solutions, Drug, Powder, Infusion, Gemcitabine, Powder for solution for infusion
Information
Domain:
Source:
Link to this page:
Please notify us if you found a problem with this document:
Documents from same domain
The Human Medicines Regulations 2012 – …
www.mhra.gov.ukThe Human Medicines Regulations 2012 – description of each Part This document provides a brief overview of each Part of the Human Medicines Regulations 2012 …
Human, Regulations, Descriptions, Medicine, 2012, Human medicines regulations 2012, Human medicines regulations 2012 description of
FULTIUM -D 800 IU CAPSULES - GOV.UK
www.mhra.gov.ukVitamin D deficiency, with levels of 25(OH)D below 25nmol/L (10ng/ml), could result in the disease rickets (or osteomalacia). The disease resulting as a deficiency is considered to be due to malabsorption
Deficiency, Capsule, Vitamin, Fultium d 800 iu capsules, Fultium, Vitamin d deficiency
GLYCOPYRRONIUM BROMIDE 0.2MG/ML …
www.mhra.gov.ukUKPAR Glycopyrronium Bromide 0.2 mg/ml Solution for Injection PL 00156/0115 1 GLYCOPYRRONIUM BROMIDE 0.2MG/ML SOLUTION FOR INJECTION (PL 00156/0115)
Solutions, Injection, 0115, Solution for injection pl 00156, 00156, Solution for injection, Pl 00156
Meloxicam - GOV.UK
www.mhra.gov.ukPL 31388/0003-4 UKPAR Alpex Pharma (UK) Ltd, Meloxicam 7.5mg and 15mg Orodispersible Tablets 3 Scientific Discussion INTRODUCTION Based on the review of the data on quality, safety and efficacy, the UK granted
PL 00010/0643 - GOV.UK
www.mhra.gov.ukUKPAR Canesten HC Cream PL 00010/0643 6 All excipients comply with their respective European Pharmacopoeia monographs, except triceteareth-4-
Public Assessment Report UKPAR Canesten Combi …
www.mhra.gov.ukThis is a summary of the Public Assessment Report (PAR) for Canesten Combi Pessary & External Cream 500mg / 2% w/w pessary & cream (PL 00010/0653).
DCPAR for Ampicillin-Salbactam 1 g--0 5 and 2 g-1 g ...
www.mhra.gov.ukPAR Ampicillin/Sulbactam 1 g/0.5 g and 2 g/1 g powder for Solution for Injection or Infusion UK/H/4322/001-2/DC 1 Public Assessment Report Decentralised Procedure
Public Assessment Report Mutual Recognition …
www.mhra.gov.ukPAR Sayana 104mg/0.65ml Suspension for Injection UK/H/0960/001/MR 1 Public Assessment Report Mutual Recognition Procedure SAYANA 104MG/0.65ML SUSPENSION FOR INJECTION
Assessment, Report, Public, Procedures, Mutual, Recognition, Public assessment report mutual recognition, Public assessment report mutual recognition procedure
Cefixime 400 mg Film-coated Tablets, PL 22805 …
www.mhra.gov.ukMHRA PAR; CEFIXIME 400 MG FILM-COATED TABLETS, PL 22805/0032-3 2 Lay summary The Medicines and Healthcare products Regulatory Agency …
Public Assessment Report Decentralised Procedure …
www.mhra.gov.ukPublic Assessment Report Decentralised Procedure LIOTHYRONINE SODIUM 20 MICROGRAM TABLETS (liothyronine sodium) Procedure No: UK/H/6516/001/DC
Assessment, Report, Public, Procedures, Public assessment report decentralised procedure, Decentralised
Related documents
Harzardous Drug Medication List *COLOR CODE AND PPE KEY
aspiruslibrary.org**COLOR CODE AND PPE KEY Drug Name Personal Protective Equiptment Required Drug Waste and Partials Empty Vials or Containers/Empty Unit Dose Packaging PPE Waste Reference
Name, Drug, Lists, Medication, Drug name, Harzardous, Harzardous drug medication list
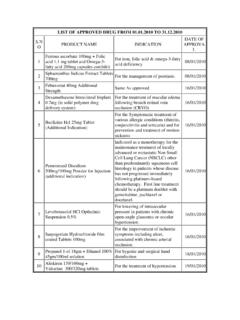
LIST OF APPROVED DRUG FROM 01.01.2010 TO 31.12
www.cdsco.nic.inLIST OF APPROVED DRUG FROM 01.01.2010 TO 31.12.2010 S.N O PRODUCT NAME INDICATION DATE OF APPROVA L 1 Ferrous ascorbate 100mg + Folic acid 1.1 mg tablet and Omega-3-
Form, Name, Drug, 2010, Approved, Of approved drug from 01, 2010 to
Prescription Drug Listing - horizonnjhealth.com
www.horizonnjhealth.comPrescription . Drug Listing. Most prescription and non-prescription (over-the-counter) drugs are covered at no cost, or low cost to you, as long as your doctor orders them and
DRUG NAME: Axitinib - BC Cancer
www.bccancer.bc.cais reported in 40% of patients on axitinib. Increased blood pressure has been reported as early as 4 days after starting axitinib, although median onset for hypertension is within the first month of treatment.
GUIDE TO EXTRAVASATION MANAGEMENT IN ADULT & …
icmwk.comGUIDE TO EXTRAVASATION MANAGEMENT IN ADUL T & PEDIATRIC PATIENTS. Large, well-designed, controlled clinical trials in humans are not available to support the
Xeloda® (capecitabine) Oral Medication Precertification ...
www.aetna.comGR-69130 (8-18) Xeloda ® (capecitabine) Oral Medication Precertification Request . Page 1 of 3 . Aetna Precertification Notification 503 Sunport Lane, Orlando, FL 32809
Aetna, Medication, Request, Precertification, Medication precertification request