Navica BinaxNow Consumer Infographi - Abbott Laboratories
BinaxNOW COVI D-19 Ag Card test are coming soon. Visit NAVICA.ABBOTT to sign up to be one of the first to know when and where the COVID-19 tests are being offered in your community. WHAT KIND OF TEST IS USED? The BinaxNOW COVID-19 Ag Card is a rapid lateral flow antigen test administered by a healthcare professional or a trained operator.
Tags:
Information
Domain:
Source:
Link to this page:
Please notify us if you found a problem with this document:
Documents from same domain
ON THE GO? TESTING THAT TRAVELS WITH YOU.
dam.abbott.comThe BinaxNOW™ COVID-19 Ag Card Home Test is a lateral flow immunoassay intended for the qualitative detection of nucleocapsid protein antigen from SARS-CoV-2. This test is authorized for prescription home use with self-collected observed direct anterior nasal (nares) swab samples from individuals aged 15 years or older who are ...
BinaxNOW™ COVID-19 Ag Card: Here's How the Test Works
dam.abbott.comsuspected of COVID-19 in the first seven days of symptoms. WHAT IS NAVICA? NAVICA is a complementary app that pairs With BinaxNOW. This first-of-its-kind app will allow people who test negative to have a temporary encrypted digital pass that displays their results, smilar to an airline boarding pass. This allows organizations to veri9 negative
A SUSTAINABLE FUTURE STARTS WITH HEALTH
dam.abbott.comFUTURE STARTS WITH HEALTH A sustainable future starts with health. It’s the foundation for all we can do in life – as individuals, families and communities. It helps societies thrive and fuels successful economies. At Abbott, sustainability means managing our company to deliver long-term impact for the people we serve – shaping the future
2020 ANNUAL REPORT - Abbott Laboratories
dam.abbott.comof Pakistan’s most pressing healthcare challenges to help people live fuller, healthier, more dignified lives. Because we believe that the best medical product is the one that helps the most people, ... us to deliver consistent growth and strong …
Panbio COVID-19 Ag Rapid Test Device - Abbott Laboratories
dam.abbott.comThe availability of a cost-effective, rapid point-of-care diagnostic test is critical to enable healthcare professionals to aid in the diagnosis of patients and prevent further spread of the virus 5. Antigen tests will play a critical role in the fight against COVID-196. Test Principle
Devices, Tests, Diagnostics, Rapid, Panbio, Covid, Panbio covid 19 ag rapid test device
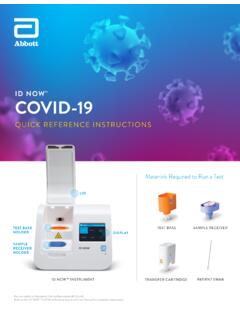
QUICK REFERENCE INSTRUCTIONS - Abbott Laboratories
dam.abbott.comFurther information can be obtained from your distributor or by contacting Technical Support: United States Europe and Middle East+ 1 855 731 2288 ts.scr@abbott.com Africa, Russia, CIS + 44 161 483 9032 EMEproductsupport@abbott.com Asia Pacific + 61 7 3363 7711 APproductsupport@abbott.com Canada + 1 800 818 8335 CANproductsupport@abbott.com IVD
Ensure Plus Advance - Abbott Laboratories
dam.abbott.comCarbohidratos g 16,80 37 Sacarosa (33%) Jarabe de maíz (62%) FOS (5%) Grasas g 4,80 11 Ácidos grasos saturados g 0,45 0,99 Ácidos grasos monoinsaturados g 2,35 5,17 Ácidos grasos poliinsaturados g 1,70 3,74 CaHMB g 0,68 1,50 Agua mg 76,50 168 Carnitina mg 18 40 Colina mg 70 154 Minerales Sodio mg 150 330 Potasio mg 270 594 Cloro mg 63 139 ...
Panbio COVID-19 Ag Rapid Test Device - Abbott Laboratories
dam.abbott.comSpecimen Collection & Extraction 1. Tilt the patient’s head back slightly about 45°-70° to straighten the passage from the front of the nose. 2. Insert the swab with a flexible shaft through the nostril parallel to the palate. Caution: Use dedicated nasopharyngeal swab for …
Devices, Tests, Collection, Specimen, Rapid, Specimen collection, Panbio, Covid, Panbio covid 19 ag rapid test device
Ensure Polvo - Abbott Laboratories
dam.abbott.comSelenio mcg 22 5,1 12 Cromo mcg 23 5,4 12 Molibdeno mcg 42 9,8 23 Vitaminas Vitamina A (palmitato) mcg RE 450 105 242 Vitamina A (β-caroteno) mcg RE 20 4,7 11 Vitamina D 3 mcg 4,8 1,1 2,6 Vitamina E mg α-TE 7,9 1,8 4,2 Vitamina K 1 mcg 33 7,7 18 Vitamina C mg 54 13 29 Ácido fólico mcg 130 30 70 Vitamina B 1 mg 0,80 0,19 0,43 Vitamina B 2 mg ...
Related documents
Abbott BinaxNOW Ag Test Card Guidance
www.maine.govFeb 12, 2021 · COVID-19 case and a negative BinaxNOW Ag Card test may work only on the day of the negative BinaxNOW result with proper personal protective equipment and symptom monitoring. (Updated 01/25/21) o A positive BinaxNOW test result indicates that the adult is a probable case. Given the
ABBOTT BINAXNOWTM COVID˜19 AG CARD TEST …
www.cdc.govAbbott BinaxNOW. TM . COVID-19 Ag Card Test Helpful Testing Tips (continued) During the Test . 15' SAMPLE CONTROL . BinaxNOW . TM . COVID -19 Ag CARD . Correct x6 Wrong . 1 . Leave the card sealed in its foil pouch until just before use and do not use the card if the card or foil pouch is open, damaged, expired or if the blue line is not present.
Tests, Testing, Tips, Card, Helpful, Abbott, Binaxnow, Covid, Abbott binaxnowtm covid 19 ag card test, Binaxnowtm, Card test helpful testing tips
Abbott BinaxNOW Overview
health.hawaii.govBINAXNOW OVERVIEW Presented by Priscilla Seabourn M.S., Ph.D. Candidate. APHL-CDC COVID-19 Associate & Joanna Kettlewell Ph.D. APHL-CDC COVID-19 Response Postdoctoral Fellow. Overview • The Binax NOW COVID-19 Ag Card is a lateral flow immunoassay intended for the qualitative detection of
Abbott BinaxNOW Rapid COVID-19 Antigen At-Home Test …
www.co.madison.oh.usAbbott BinaxNOW Rapid COVID-19 Antigen At-Home Test Kit This test kit must be used with a certified proctor using your smartphone or computer with a camera and microphone. The entire process only takes 20 minutes. For instructions visit howtotest.madisonph.org or scan the QR code. This test kit is authorized for people age 4 and older.
BinaxNOW COVID-19 Antigen Self-Test Kit Instructions
henrycohd.orgJan 01, 1990 · BinaxNOW COVID-19 Self-Test FAST Results Minutes . ENTER YOUR RESULTS Your test result will be shared with your connected organizations as well as public health authorities to meet COVID-19 reporting requirements. Name: Date of Birth: Positive Negative Invalid CANCEL SUBMIT Maggie Wielinski 09/04/1983 .