POLICIES AND PROCEDURES MANUAL CLIA #01D0665512
Laboratory personnel will monitor test requests for appropriateness to the patient’s age, sex, and diagnosis. If any test request or result appears inappropriate, proper consultation should be obtained. H. Personnel Assessment An ongoing evaluation of all testing personnel will be conducted through use of
Tags:
Manual, Personnel, Policies, Procedures, Clia, Policies and procedures manual clia, 01d0665512
Information
Domain:
Source:
Link to this page:
Please notify us if you found a problem with this document:
Documents from same domain
Alabama Department of Public Health Central Office ...
www.alabamapublichealth.govNursing Home Unit, Lisa Pezent, B.S.N. 700 206-5111 CLIA Laboratory Unit, Patricia Watson, B.S., M.T. (ASCP) 700 206-5120 TO FILE A COMPLAINT Against an Assisted Living Facility 1-866-873-0366 TO FILE A COMPLAINT Against a Nursing Home/Skilled Nursing Facility 1 …
Practical Tips on Faculty Management of Patients with ...
www.alabamapublichealth.govPractical Tips on Management of Patients with Dementia in the Home Satellite Conference and Live Webcast Wednesday, May 16, 2007 2:00 - 4:00 p.m. (Central Time) Produced by the Alabama Department of Public Health Video Communications and Distance Learning Division Faculty ... Aphasia …
ICD·9·CM Casefinding Code List for Reportable Tumors
www.alabamapublichealth.govICD·9·CM Casefinding Code List for Reportable Tumors ADPH|ASCR Last Update 3/24/2016 Page 3 Supplemental Codes 042 AIDS with specified malignant neoplasms
Code, Lists, Reportable, 183 9, 183 cm casefinding code list for reportable, Casefinding
FREQUENTLY ASKED QUESTIONS REGARDING THE …
www.alabamapublichealth.govA. The law provides that the date of the marriage is the that the affidavit date on the Alabama Marriage Certificate form is signed by the two spouses, so long as the completed and notarized form is provided to the probate office for recording within 30 days of …
Form, Question, Affidavits, Frequently, Asked, Regarding, Notarized, Frequently asked questions regarding the, Notarized form
Chapter 420 -5-19/Appendices Health APPENDIX II
www.alabamapublichealth.govHealth Chapter 420-5-19/Appendices Supp. 12/31/16 A-4 Section IV. Surrogate Consent. I, the undersigned, am the surrogate certified to make decisions, in consultation with the attending physician, regarding the providing, withholding, or withdrawal of life-sustaining treatment for the patient/resident.
Montgomery County Food Inspection Scores Updated 03/16/20
www.alabamapublichealth.govCaporal Mexican Restaurant 6132 Atlanta Highway 12/09/19 96 Capri Theatre 1045 East Fairview Avenue 03/06/20 98 Captain D's 10640 Chantilly Parkway 03/02/20 92 Captain D's #3327 2674 Zelda Road 01/17/20 97. Montgomery County Food Inspection Scores ... Montgomery County Food Inspection Scores ...
Human Resources Procedures Manual
www.alabamapublichealth.govThe Human Resources Procedures Manual (Manual) has been developed by the Office of Human Resources (HR). The purpose of the Manual is …
Manual, Human, Procedures, Resource, Human resources procedures manual
USE ONLY FOR A VITAL EVENT WHICH OCCURRED IN …
www.alabamapublichealth.govUSE ONLY FOR A VITAL EVENT WHICH OCCURRED IN ALABAMA . The fee for a birth, death, marriage or divorce record search is $15.00, which includes the cost of one certified copy OR a Certificate of Failure to Find. For additional copies of the same record ordered at the same time, the fee is $6.00 each. Amendments, adoptions, legitimations, and
Promoting Physical Activity for Infants and Toddlers in EC ...
www.alabamapublichealth.govLearning Outcomes At the end of this session, participants will be able to: • Acknowledge the importance of daily physical activity for infants and toddlers. • Describe developmentally appropriate activities to promote infant growth and development. • Give examples of appropriate indoor and outdoor physical activities for toddlers.
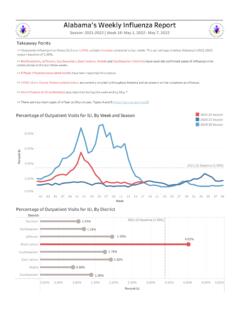
Alabama's Weekly Influenza Report
www.alabamapublichealth.gov=> COVID-19, Respiratory syncytial virus, and Human rhinovirus/enterovirus are currently circulating throughout Alabama and can present similar symptoms as influenza. => No influenza or ILI outbreak(s) were reported during the week ending December 25.
Report, Alabama, Weekly, Rhinovirus, Influenza, Alabama s weekly influenza report
Related documents
S&C-16-18-CLIA Personnel Policies for Individuals ...
www.cms.govS&C-16-18-CLIA Personnel Policies for Individuals Directing or Performing Non-waived Tests . Frequently Asked Questions . Q1. What is Primary Source Verification (PSV)? A1. PSV is the process of confirming an individual’s credentials by verifying that a degree, certificate, or diploma was received; that licenses were granted; and, by ...
CMS-209 Laboratory Personnel Report (CLIA)
www.cms.govlaboratory personnel report (clia) (for moderate and high complexity testing) form approved omb no. 0938-0151 department of health and human services centers for medicare & medicaid services 1. laboratory name 2. clia identification number 3. laboratory address (number and street) city state zip code 4. ...
Services, Report, Laboratory, Center, Medicare, Medicaid, Personnel, Clia, Centers for medicare amp medicaid services, Laboratory personnel report
A Practical Approach to CLIA Requirements and AABB, CAP ...
aabb.confex.comOct 15, 2018 · CLIA Brochure #10 What Do I Need to Do to Assess Personnel Competency? Competency assessment, which includes the six procedures, must be performed for testing personnel for each test that the individual is approved by the laboratory director to perform.
Practical, Requirements, Approach, Personnel, Clia, Practical approach to clia requirements
POLICIES AND PROCEDURES MANUAL CLIA #01D0665512
www.adph.orgduring a CLIA inspection. 2. New testing personnel must have competency twice during their first year of training, then annually thereafter. 3. Nonwaived workshop trainings are presented twice a year at the Bureau of Clinical Laboratories. E. Proficiency Testing ACHDLS will participate in a CMS-approved PT program. PT results will be reviewed
Manual, Personnel, Policies, Procedures, Clia, Policies and procedures manual clia, 01d0665512
CAP Personnel Requirements by Testing Complexity
documents.cap.orgJul 18, 2016 · While CLIA may allow for non-physician or nondoctoral degreed individuals to direct Waived and Moderate Complexity laboratories, CAP does not. 1 The license to practice medicine must be in the jurisdiction where the laboratory is located(if required). Military and VA personnel may be licensed in any US State. Page 2 of 3 OPM 6918 June 2016
Requirements, Personnel, Complexity, Clia, Personnel requirements
POLICIES AND PROCEDURES MANUAL CLIA #01D0665512
www.adph.orgduring a CLIA inspection. 2. New testing personnel must have competency twice during their first year of training, then annually thereafter. 3. Nonwaived workshop trainings are presented twice a year at the Bureau of Clinical Laboratories. E. Proficiency Testing ACHDLS will participate in a CMS-approved PT program. PT results will be reviewed
READY? SET? TEST!
www.cdc.govCLIA requires that waived tests must be simple and have a low risk for an incorrect result. However, this does not mean waived tests are completely error-proof. To decrease the likelihood of incorrect results, waived testing needs to be performed correctly, by trained personnel and in an environment where good testing practices are followed.
LabGuide 16 Personnel Training & Competency Assessment
labflorida.comPersonnel training must be documented and retained in the personnel files of all laboratory testing personnel. Competency Assessment Competency Assessment is the means to confirm that training is effective and that personnel are capable of following established procedures to accurately perform
Frequently Asked Questions
www.azdhs.govthe State Agency and, once processed, the facilitywill receive an invoice (CLIA user fee coupon) from CMS in Maryland. Once the invoice is paid, CMS will mail the facility their CLIA certificate. The entire application process usually takes 4-6 weeks. …
Question, Frequently, Asked, Frequently asked questions, Clia
PROVIDER- PERFORMED MICROSCOPY PROCEDURES
www.cdc.govother point-of-care location. The Clinical Laboratory Improvement Amendments of 1988 (CLIA) Certificate for Provider-Performed Microscopy (PPM) procedures issued by the Centers for Medicare & Medicaid Services (CMS) permits a laboratory or testing site to perform a limited list of moderate complexity microscopic tests, as well as any waived tests.
Laboratory, Clinical, Improvement, Procedures, Provider, Amendment, Clia, Microscopy, Clinical laboratory improvement amendments, Performed, Provider performed microscopy procedures
Related search queries
CLIA Personnel, Laboratory personnel report, CLIA, Centers for medicare & medicaid services, Practical Approach to CLIA Requirements, Personnel, POLICIES AND PROCEDURES MANUAL CLIA #01D0665512, Personnel Requirements, Complexity, READY? SET? TEST, Frequently Asked Questions, PROVIDER- PERFORMED MICROSCOPY PROCEDURES, Clinical Laboratory Improvement Amendments