Rare Pediatric Disease Priority Review Vouchers, Draft ...
Rare Pediatric Disease Priority Review Vouchers, Guidance for Industry . DRAFT GUIDANCE . This guidance document is being distributed for comment purposes only.
Tags:
Review, Disease, Pediatric, Rear, Priority, Voucher, Rare pediatric disease priority review vouchers
Information
Domain:
Source:
Link to this page:
Please notify us if you found a problem with this document:
Documents from same domain
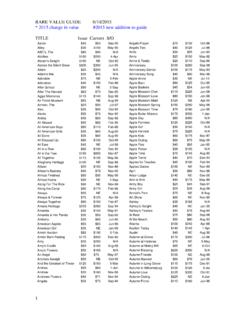
eSubmitter Quick Guide - Food and Drug …
www.fda.govAllows you to create a new submission entry. The New Submission Data Dialog box will ap-pear. See section Creating a New Submission for more detailed information.
Guide, Information, Drug, Food, Food and drug, Quick, Esubmitter quick guide, Esubmitter
1 APPENDIX B - PRODUCT NAME SORTED BY …
www.fda.govjune 2018 - approved drug product list b - 2 appendix b - product name sorted by applicant ** a ** * abbvie inc norvir, ritonavir technivie, ombitasvir
Product, Name, Appendix, Applicants, Appendix b product name sorted by, Sorted, Appendix b product name sorted by applicant
Study Data Specifications - Food and Drug …
www.fda.govStudy Data Specifications STUDY DATA SPECIFICATIONS . These specifications are for submitting animal and human study datasets in electronic format.
Study, Data, Specification, Drug, Food, Food and drug, Study data specifications, Study data specifications study data specifications
Sanofi Pasteur 20 April 2016v0.2 095 - ActHIB LE6940
www.fda.govSanofi Pasteur 20 April 2016v0.2 095 - ActHIB® LE6940 Confidential/Propietary Information Page 2 of 19 1 . FULL PRESCRIBING INFORMATION: 2 . …
April, Sanofi pasteur 20 april 2016v0, Sanofi, Pasteur, 2016v0, 2 095
Repackaging of Certain Human Drug Products by …
www.fda.govRepackaging of Certain Human Drug Products by Pharmacies and Outsourcing Facilities. Guidance for Industry . Additional copies are available from: Office of …
Product, Drug, Human, Certain, Repackaging of certain human drug products by, Repackaging, Repackaging of certain human drug products by pharmacies, Pharmacies
POLICY AND PROCEDURES OFFICE OF …
www.fda.govMANUAL OF POLICIES AND PROCEDURES CENTER FOR DRUG EVALUATION AND RESEARCH MAPP 5040.1 Originating Office: Office of Pharmaceutical Quality Effective Date: 5/24/2004; 2/26/2014, 01/26/2017 Page 1 of 9
Manual, Pharmaceutical, Quality, Office, Office of, Office of pharmaceutical quality
BIORESEARCH MONITORING TECHNICAL …
www.fda.govBIORESEARCH MONITORING TECHNICAL CONFORMANCE GUIDE Containing Technical Specifications For questions regarding …
Guidance for Industry - Food and Drug …
www.fda.govGuidance for Industry . Recommendations for Screening, Testing, and Management of Blood Donors and Blood and Blood Components Based on Screening
TABLE OF CONTENTS - Food and Drug …
www.fda.govList of Abbreviations Used . General Abbreviations . AR Antimicrobial Resistance . BAP Blood Agar Plate . CCA Campy-Cefex Agar Plate . CDC Centers for …
Drug, Food, Food and drug, Content, Table of contents, Table
148 COAGULA BeneF D RECOMBINANT W IX TION …
www.fda.govprocedures was 85 ± , Coagulation Factor IX (Recombinant), is indicated for the control and ). Patients with factor IX inhibitors may be at an increased risk of
Recombinant, Coagulation, 148 coagula benef d recombinant w, Coagula, Benef
Related documents
Tennessee Natural Heritage Program Rare Species ...
madisonscd.comTennessee Natural Heritage Program Rare Species Observations For Tennessee Counties If you need assistance using the lists or interpreting data, feel free to contact us:
Programs, Species, Tennessee, Rear, Observation, Heritage, Counties, Heritage program rare species observations for tennessee counties
New York Rare Plant Status Lists November 2017
www.dec.ny.govNew York Rare Plant Status List May 2003 Edited by Stephen M. Young and Troy W. Weldy New York Natural Heritage Program A Partnership between SUNY …
M M M M - Medium Rare
www.mediumrarerestaurant.comvin rouge Red Wine vin blanc White Wine vin mousseux Sparkling Wine Fresh Pinot Grigio Italy $9 $34 frais Annalisa Crisp Sauvignon Blanc France $10 $37
Final Agenda Welcome of (ORDR) by
events-support.com10:45 a.m. NIH Director Remarks Francis S. Collins, M.D., Ph.D., Director, NIH 11:00 a.m. Session 2: There is Power in Numbers — Harnessing Patient Data through Registries An essential step in improving the diagnosis and treatment of a rare disease is collecting enough
RARE VALUE GUIDE 8/10/2015 - P. Buckley Moss
www.pbuckleymoss.comRARE VALUE GUIDE 8/10/2015 * 2015 change in value #2015 new addition to guide TITLE Issue Current S/O 3 Bringing in the Apples $180 $450 Aug-94
STANDARD SPECIFICATIONS FOR PERMANENT MAGNET …
www.intemag.commmpa standard no. 0100-00 standard specifications for permanent magnet materials magnetic materials producers association 8 south michigan anenue • suite 1000 • chicago, illinois 60603
Standards, Specification, Permanent, Magnet, Standard specifications for permanent magnet