Search results with tag "Recombinant"
Role of the Recombinant DNA Advisory Committee and the ...
research.tu.eduNIH Recombinant DNA Advisory Committee (RAC)Advisory Committee (RAC) Federal advisory committee providing advice and recommendations to the NIH Director regarding recombinant DNA research Unique public forum for the discussion of science, safety, and ethics of recombinant DNA research Ri d l liil t fReviews and analyzes clinical gene transfer
Genetic Engineering / Recombinant DNA technology
microrao.comGenetic Engineering / Recombinant DNA technology Genetic engineering is a broad term referring to manipulation of an organisms’ nucleic acid. Organisms whose genes have been artificially altered for a desired affect is often called genetically modified organism (GMO). Recombinant DNA technology (rDNA) is technology that is used to cut a known DNA
Use of Recombinant Zoster Vaccine in Immunocompromised ...
www.cdc.govZoster Vaccine Recombinant, Adjuvanted (Shingrix, GlaxoSmithKline [GSK]) is a 2-dose (0.5 mL each) subunit . vaccine containing recombinant glycoprotein E in combination with adjuvant (AS01. B) that was licensed in the United States . for prevention of herpes zoster for adults aged ≥50 years by the Food and Drug Administration (FDA) and ...
INTRODUCTION WHAT IS A VACCINE? WHAT ARE THE ... - IJSER
www.ijser.org5. What is a Recombinant vaccine? Vaccine produced by using recombinant DNA technology (i.e. mixing of two DNA from different sources) is called recombinant vaccine. This involves inserting the DNA encoding antigen (such as bacterial surface protein) thatstimulates an immune response into bacterial or mammalian
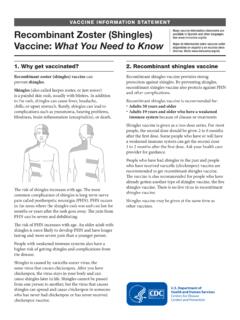
Vaccine Information Statement: Recombinant Zoster ...
www.cdc.govRecombinant Zoster (Shingles) Vaccine: What You Need to Know 1. Why get vaccinated? Recombinant zoster (shingles) vaccine can prevent shingles. Shingles (also called herpes zoster, or just zoster) is a painful skin rash, usually with blisters. In addition to the rash, shingles can cause fever, headache, chills, or upset stomach.
HOME recombinant murine cytokines - ImmunoTools
www.immunotools.deHOME recombinant murine cytokines rm CXCL12b (Stromal Cell-Derived Factor-1 beta / SDF-1beta) Catalog# Biol.Activity Size Price
Growth and maintenance of insect cell lines
tools.thermofisher.comof protein expression. Once you have confirmed expression of your recombinant protein, other cell lines can be tried for optimization of expression levels. Design guidelines . When designing a scheme for purification of polyhistidine-tagged recombinant proteins, note that serum-free media cannot be applied directly to a metal-chelating
PRECLINICAL SAFETY EVALUATION OF BIOTECHNOLOGY …
database.ich.orgrecombinant plasma factors, growth factors, fusion proteins, enzymes, receptors, hormones, and monoclonal antibodies. The principles outlined in this guidance may also be applicable to recombinant DNA protein vaccines, chemically synthesised peptides, plasma derived products,
The Gelsinger Case - University of Alabama at Birmingham
www.uab.eduThe Recombinant DNA Advisory Committee (RAC) at the National Institutes of Health approves a clinical protocol from the Institute for Human Gene Therapy, "Recombinant Adenovirus Gene Transfer in Adults With Partial Ornithine Transcarbamylase rOTC] Deficiency." The principal investigator is Steven E. Raper, also of the University of Pennsylvania.
Biotechnology and Recombinant DNA
www2.sunysuffolk.eduBiotechnology and Recombinant DNA (Chapter 9) Lecture Materials for Amy Warenda Czura, Ph.D. Suffolk County Community College Eastern Campus Primary Source for Þgures and content:
HOME recombinant human cytokines - ImmunoTools
www.immunotools.deHOME recombinant human cytokines rh BMP-7 HEK (Bone Morphogenetic protein-7 HEK) Catalog# Biol.Activity Size Price
TD : clonage/ADN recombinant - INRA
www1.montpellier.inra.frLes stratégies de clonage: 1. Sélectionner les clones recombinants Insertion dans la partie LacZa de l’opéron lactose Le 5-bromo-4-chloro-3-indolyl-b-D galactoside (X-Gal) est clivé par la β-galactosidase pour donner une substance bleue insoluble et en présence d’IPTG (colonies bleues) Si on introduit un ADN étranger dans le
Antihemophilic Factor, Recombinant only
www.abopharmaceuticals.com1 ReFacto Antihemophilic Factor, Recombinant only This product’s label may have been revised after this insert was used in production. For further product information and current package
HEPATITIS B VACCINE (RECOMBINANT) - CTVIA
www.ctvia.orgRECOMBIVAX HB Hepatitis B Vaccine (Recombinant) 7994321 2 CLINICAL PHARMACOLOGY Hepatitis B virus is one of several hepatitis viruses that cause a systemic infection, with a
The Structure and Function of ... - Biochemical Society
www.biochemistry.orgin the area of genetic engineering and recombinant DNA technology (which are covered in Booklet 7 of the BASC series) have only been possible as a result of detailed understanding of the structure of DNA and RNA. It is therefore not surprising to see nucleic acids included in the compulsory core subject matter of all of the linear and modular A-
Studies on recombinant lipase production by E. Coli ...
medcraveonline.comE. Coli: effect of media and bacterial expression system optimization. () ...
HIGHLIGHTS OF PRESCRIBING INFORMATION …
www.fda.govFULL PRESCRIBING INFORMATION 1 INDICATIONS AND USAGE XYNTHA,Antihemophilic Factor (Recombinant), is indicated for usein adults and children with hemophilia A(congenital factor VIII deficiency)for:
Guidelines on the quality, safety, and efficacy of ...
www.who.intmanufacturers with guidance on the quality, safety and efficacy of biotherapeutic protein products prepared by recombinant deoxyribonucleic acid (DNA) technology (rDNA-derived biotherapeutics) and intended for use in humans. The guidelines are based on experience gained over three decades in this technically demanding field and replace
Annex 3 - WHO
www.who.intWHO Expert Committee on Biological Standardization Sixty-first report Recommendations published by WHO are intended to be scientific and advisory. Each of the following sections constitutes guidance for national regulatory authorities (NRAs) and for manufacturers of ... rcDNA residual cellular DNA rDNA recombinant DNA REO3 reovirus 3 RFLP ...
Biochemistry and Molecular Biology - kau
www.kau.edu.saCover illustration Main image Electrophoresis gel showing recombinant protein. Photographer: J.C. Revy. Courtesy of Science Photo Library. Top inset Transcription factor and DNA molecule.
Guidelines on the nonclinical evaluation of vaccine adjuvants …
www.who.intimmunostimulatory moieties that are engineered by recombinant DNA technology to be an inherent part of the antigen molecule (e.g., fusion proteins) or the immunogen (e.g., vectored vaccines). In this context, it should be noted that no vaccine adjuvant is authorized in its own right, but only as a component of a particular adjuvanted vaccine.
COVID-19 Vaccine AstraZeneca - Embolic and thrombotic ...
www.ema.europa.euVaccine (ChAdOx1-S [recombinant]) – COVID-19 Vaccine AstraZeneca (Other viral vaccines) EMA/PRAC/157045/2021 Page 5/50 artery thrombosis, cerebral venous sinus thrombosis. Other reported events included pulmonary embolism (8), thrombocytopenia (6), disseminated intravascular coagulation (4), deep vein thrombosis
Guideline on Live Recombinant Viral Vectored Vaccines
www.ema.europa.euAlthough vaccines containing live-attenuated viruses are typically not adjuvanted, guidance on the co-use of immunomodulators is provided by the guideline on Adjuvants in Vaccines for Human Use (CHMP/VEG/134716/04)[3] and by the accompanying Explanatory Note (CHMP/VWP/244894/2006)[4]. 4. Legal basis
Vaccine Information Statement: Inactivated Influenza Vaccine
www.immunize.orgVaccine Information Statement • Influenza, Inactivated (8/6/21)• Arabic Translation • Distributed by the Immunization Action Coalition Influenza (inactivated or recombinant) VIS – Arabic (8/6/21) عم ازنولفنلإا ميعطت نوذخأي نيذلا راغصلا لافطلأا حبصي دقو ...
Meningococcal B Multicomponent - Alberta Health Services
albertahealthservices.ca5. GlaxoSmithKline Inc. (2019, February 25) Bexsero® Multicomponent meningococcal B vaccine (recombinant, adsorbed). Product Monograph. 6. National Advisory Committee on Immunization (2014 April) Advice for the Use of Multicomponent Meningococcal Serogroup B …
Protocole d’utilisation thérapeutique et de recueil de ...
ansm.sante.frLe tixagévimab et le/ Cilgavimab sont deux anticorps monoclonaux recombinants humains IgG1k, avec des substitutions d’acides aminées pour prolonger la demi-vie des anticorps (YTE) et réduire leur fonction effectrice et le risque potentiel de renforcement de …
Application of Genetic Engineering
epgp.inflibnet.ac.inIn practice, since DNA contains the genes to build certain proteins, by changing the DNA sequence, ... The isolation and preparation of DNA fragments 2. Production of recombinant by joining the fragments to a suitable vector ... incompatibility problems can be avoided by using eukaryotic cells as hosts. The use of eukaryotic host
Exercice 1 - sorbonne-universite.fr
step.ipgp.jussieu.fra- Proposez un protocole de clonage et indiquez comment vous sélectionnez les clones recombinants. ! Préparation de l'insert: Extraction d'ADN génomique de la bactérie. Digestion partielle de cet ADN par l'enzyme de restriction Eco RI, de manière à …
Applications for DNA, RNA, and Protein Analysis
www.agilent.com– The Protein 80 and Protein 230 kits provide fast and easy analysis of a wide range of samples, whether expressed recombinant proteins, purified proteins, stability studies, or an antibody quality check. Microfluidic electrophoresis provides size, purity, and concentration information for 10 protein samples in less than 30 minutes.
CRISPR 101
crispr.worksrestriction enzymes in concert with recombinant adeno-associated viruses (rAAVs). AAV is a non-pathogenic virus ... endonuclease protein (Cas9). The Cas9 protein functions as a pair of molecular ... the PAM for the most widely used Cas9 from Streptococcus pyogenes is 5′-NGG-3′, where N is any nucleotide. In its natural form, the gRNA ...
pcDNA™3.1(+) pcDNA™3.1(–) - tools.thermofisher.com
tools.thermofisher.comPurification Kit 100 preps K2100-01 ... Test for expression of your recombinant gene by western blot analysis or functional assay. 2 Cloning into pcDNA™3.1 Introduction Diagrams are provided on pages 3-4 to help you design a cloning strategy for
Analysis of SARS-CoV-2 (COVID-19) Vaccine Candidates
s27589.pcdn.coS protein expression Vero monkey cells Hörner et al., PNAS 22Dec2020 Hörner et al. Supplement Israel Institute for Biological Research (IIBR) Israel Replication-competent recombinant vesicular stomatitis virus (VSVΔG) “IIBR-100” Given: Intramuscular 1 dose Phase 1/2 BHK hamster cells Vero monkey cells Yahalom-Ronen et al., bioRxiv 19June2020
Cosmetic Formulation - ANME
www.anme.com.mxmicroorganisms where genes are modified by recombinant DNA technologies. But regardless how great the science and the medical proofs behind a new product, bad or just indifferent aesthetics can hurt the performance in the marketplace. New cosmetic formulations usually are the result of systematic development
MANAGEMENT AND DISPOSAL OF BIOLOGICAL WASTE
www.tamiu.edurecombinant DNA biotechnology and genetic manipulation. Definitions of other terms used in this document can be found in APPENDIX A. Biohazardous waste generated at Texas A&M International University is treated by thermal or chemical disinfection, by encapsulation (solidification), or by incineration.
乾燥組換え帯状疱疹ワクチン シングリックス 筋注用 主要な …
www.mhlw.go.jpDNA Nucleocapsid Tegument ... ACIP(The Advisory Committee on Immunization Practices ... RZV= Recombinant Zoster Vaccine ZVL= Zoster vaccine Live 3. 一般名 乾燥組換え帯状疱疹ワクチン(チャイニーズハムスター卵巣細胞由来) 乾燥弱毒生水痘ワクチン ...
HISTORY OF DRUG DISCOVERY AND DEVELOPMENT
scalettar.physics.ucdavis.eduments for gynecological problems. Medications were based mainly on herbal products such as myrrh, frankincense, castor oil, fennel, sienna, thyme, linseed, ... practice of the Greeks. Dioscorides and Galen were two noted physicians in ... development of recombinant DNA products utilizing
Vaccines Made from Genetically Modified Organisms
www.ams.usda.govNov 29, 2011 · 128 recombinant DNA technology (including gene deletion, gene doubling, introducing a foreign gene, ... 154 disease or health problems occur, or may occur, and no alternative permitted treatment or management 155 practice exists, or, in cases required by law, vaccination of livestock, the use of parasiticides, or therapeutic ...
Hepatitis B vaccines for Australians - fact sheet
www.tri.edu.auThe hepatitis B-containing vaccines registered in Australia are subunit vaccines that contain HBsAg, produced by recombinant DNA technology from yeast cells. These vaccines are available either as monovalent antigen formulations or in combination with other vaccine antigens. Monovalent hepatitis B vaccine comes in paediatric
Recombinant Protein Expression & Purification ...
www.genscript.comProtein Expression as Inclusion Bodies 16 What is an inclusion body? • When E.coli is transformed to manufacture large amounts of recombinant protein, the protein sometimes forms dense aggregates of insoluble misfolded proteins, known as inclusion bodies. Benefit • allow high protein concentrations • protect sensitive proteins from
Recombinant protein vaccines produced in insect cells
www.who.intMeasles Measles virus (genus Morbillivirus, family Paramyxoviridae) H and F proteins H, F, and N viral proteins Rubella Rubella virus (= togavirus of the genus Rubivirus) E1 HPV Human papilloma virus (>100 subtypes) L1 structural protein Japanese Encephalitis JapaneseEncephalitis (JE) virus (Flaviviridae) Glycoprotein E
Similar queries
The Recombinant DNA Advisory Committee and, Recombinant DNA Advisory Committee, Advisory Committee, Recombinant DNA, Genetic Engineering / Recombinant DNA technology, Genetic Engineering / Recombinant DNA technology Genetic engineering, Recombinant, Vaccine Information Statement, Shingles, HOME recombinant murine cytokines, Protein Expression, Expression, Recombinant protein, PRECLINICAL SAFETY EVALUATION OF BIOTECHNOLOGY, Protein, The Gelsinger Case, University of Alabama at Birmingham, The Recombinant DNA Advisory Committee, Biotechnology and Recombinant DNA, HOME recombinant human cytokines, Recombinants, Antihemophilic Factor, Recombinant only, Hepatitis B Vaccine Recombinant, Hepatitis B, Hepatitis, Infection, Recombinant DNA technology, And bacterial expression system optimization, HIGHLIGHTS OF PRESCRIBING INFORMATION, Antihemophilic Factor, Factor, Efficacy of biotherapeutic protein products prepared by recombinant, Annex 3, Committee, Advisory, Biochemistry and Molecular Biology, Vaccine, Live Recombinant Viral Vectored Vaccines, Adjuvanted, Immunization Action Coalition, Practice, Problems, Comment, Applications for DNA, RNA, and Protein Analysis, CRISPR, Cas9, Cas9 protein, Streptococcus pyogenes, Purification, Texas A&M International University, Hepatitis B vaccine, Recombinant Protein Expression, Recombinant protein vaccines produced in insect, Virus, Human papilloma virus