Search results with tag "Adjuvanted"
Guidelines on the nonclinical evaluation of vaccine ...
www.who.intadjuvanted vaccines, to review the draft guidelines prepared by the drafting group and to seek consensus on key regulatory issues. The approaches to nonclinical and initial clinical evaluation of vaccine adjuvants and adjuvanted vaccines discussed in this document are a result of the efforts of this and other international working groups. Scope
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS
www.ema.europa.euHerpes zoster vaccine (recombinant, adjuvanted) 2. QUALITATIVE AND QUANTITATIVE COMPOSITION . After reconstitution, one dose (0.5 mL) contains: Varicella Zoster Virus. 1. glycoprotein E antigen. 2,3. 50 micrograms . 1 Varicella Zoster Virus = VZV . 2 adjuvanted with AS01 B containing: plant extract . Quillaja saponaria. Molina, fraction 21 (QS ...
FIRST DRAFT Guideline on clinical evaluation of vaccines REV1
www.ema.europa.eu118 unlicensed) vaccines, adjuvanted or non-adjuvanted, and to support modifications to vaccines in the 119 post-approval period (e.g. changes in, or additions to, the posology, the age range for use or 120 recommendations for concomitant vaccination). 121 The guidance addresses trials to document vaccine safety, immunogenicity and/or efficacy.
Module 2.6: Nonclinical Written and Tabulated Summary 2.6 ...
www.mhlw.go.jpGSK™s AS03-adjuvanted H5N1 influenza vaccine is a monovalent, split-virion, inactivated, adjuvanted vaccine. The influenza strain contained in the vaccine is A/H5N1/Indonesia/5/2005, prepared by reverse genetics. The antigen is presented in a 10mL Type I glass vial (10 doses) and the AS03 adjuvant presented in a 3mL Type I glass vial (10 doses).
Guideline on Live Recombinant Viral Vectored Vaccines
www.ema.europa.euAlthough vaccines containing live-attenuated viruses are typically not adjuvanted, guidance on the co-use of immunomodulators is provided by the guideline on Adjuvants in Vaccines for Human Use (CHMP/VEG/134716/04)[3] and by the accompanying Explanatory Note (CHMP/VWP/244894/2006)[4]. 4. Legal basis
COVID-19 Clinical Working Group Summary Document Topic ...
media.tghn.orgmay contribute more to the efficacy of adenoviral and mRNA than protein-based vaccines, including adjuvanted subunit and nanoparticle vaccines. The complex, composite nature of protection, as captured in the equation, is further illustrated by (i) the early onset of protection against symptomatic COVID-19 (i.e., 10 days after the first
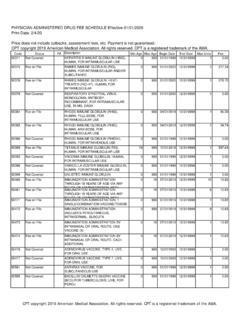
PHYSICIAN ADMINISTERED DRUG FEE SCHEDULE Effective …
www.medicaid.ms.govadjuvanted preservative free 0.5 mg dosage for intramuscular use 0 999: 01/01/2020 12/31/9999: 1 0.00: 90698 fee on file: diphtheria, tetanus toxoids, acellular pertussis vaccine, haemophilus influenzae type b, and inactivated poliovirus vaccine, for intramuscular use 0 18: 08/01/2008 12/31/9999: 1 0.00: 90700 fee on file: diphtheria, tetanus ...
Evidence Assessment: Sinopharm/BBIBP COVID-19 vaccine
cdn.who.intApr 02, 2021 · • 2-dose β-propiolactone-inactivated, aluminiumhydroxide-adjuvanted COVID-19 vaccine administered on a 0/21-28-day schedule for the prevention of COVID-19 disease • Authorized by the China National Medical Products Administration on December 31, 2020 • Authorized by 45 countries/jurisdictions for use in adults ≥18 years
Category I Vaccine Long Code Descriptors
www.ama-assn.org90739 Hepatitis B vaccine (HepB), CpG-adjuvanted, adult dosage, 2 dose or 4 dose schedule, for intramuscular use December 30, 2021 July 1, 2022 CPT® 2023 # ⚫90759 Hepatitis B vaccine (HepB), 3-antigen (S, Pre-S1, Pre-S2), 10 mcg dosage, 3 dose schedule, for intramuscular use December 30, 2021 July 1, 2022 CPT® 2023
Influenza Vaccine Products for the 2021-2022 Influenza …
www.immunize.orgaIIV4 = adjuvanted quadrivalent inactivated influenza vaccine. 2. An administration code should always be reported in addition to the vaccine product code. Note: Third party payers may have specific policies and guidelines that might require providing additional information on their claim forms. 3. Dosing for infants and children age 6 through
The COVID-19 Vaccine Development Landscape - CGMH
www1.cgmh.org.twto develop adjuvanted vaccines against COVID-19, and vaccine developers including GlaxoSmithKline, Seqirus and Dynavax have committed to making licensed adjuvants (AS03, MF59 and CpG 1018, respectively) available for use with novel COVID-19 vaccines developed by others. Public information on the specific SARS-CoV-2 antigen(s) used in vaccine
Guideline on Influenza Vaccines
www.ema.europa.euSpecific requirements for adjuvanted vaccines or live attenuated vaccines are exemplified in dedicated paragraphs as appropriate. For further details, this section should be c omplemented with the principles outlined in the guidelines listed in section 3. Additionally the WHO Guideline on non-clinical evaluation of vaccines (WHO Technical
Vaccination (Immunization) – Medicare Advantage Policy ...
www.uhcprovider.comVaccines listed below are eligible for Medicare Part B payment. These vaccines may be reimbursed regardless of the setting in which they are furnished. ... inactivated (IIV), subunit, adjuvanted, for intramuscular use (FDA approved for adults 65 years of age and older) 90654 . Influenza virus vaccine, trivalent (IIV3), split virus ...
Regulatory Considerations in the Safety Assessment of ...
www.fda.govRegulatory Considerations in the Safety Assessment of Adjuvants and Adjuvanted Preventive Vaccines . SLIDE 1 . This talk will cover the Regulatory Considerations in the Safety Assessment of
Guidelines on the nonclinical evaluation of vaccine ...
www.who.intPage 5 Introduction This document provides guidance to national regulatory authorities (NRAs) and manufacturers on the nonclinical and initial clinical evaluation of vaccine adjuvants and adjuvanted vaccines …
Similar queries
Vaccine, Adjuvanted vaccines, Vaccine adjuvants and adjuvanted vaccines, SUMMARY OF PRODUCT CHARACTERISTICS, Adjuvanted, Vaccines, Live Recombinant Viral Vectored Vaccines, Influenza Vaccine Products, Adjuvants, On Influenza Vaccines, Evaluation, Regulatory Considerations in the Safety, Adjuvants and Adjuvanted Preventive Vaccines, Guidelines on the nonclinical evaluation