Reference ID: 3433133
Genital Mycotic Infections 5.5. Increases in Low-Density Lipoprotein Cholesterol (LDL-C) 5.6. Bladder Cancer 5.7. Macrovascular Outcomes 6 ADVERSE REACTIONS . 6.1 Clinical Trials Experience 8 USE IN SPECIFIC POPULATIONS 8.1 Pregnancy 8.3. Nursing Mothers 8.4. Pediatric Use 8.5. Geriatric Use 8.6. Renal Impairment 8.7. Hepatic Impairment 10 ...
Tags:
Information
Domain:
Source:
Link to this page:
Please notify us if you found a problem with this document:
Documents from same domain
HIGHLIGHTS OF PRESCRIBING INFORMATION …
www.accessdata.fda.govweakness, diplopia, ptosis, dysphagia, dysphonia, dysarthria, urinary incontinence and breathing difficulties. These symptoms . FULL …
Effexor (venlafaxine hydrochloride) Tablets …
www.accessdata.fda.govEffexor ® (venlafaxine hydrochloride) Tablets Rx only . Suicidality and Antidepressant Drugs Antidepressants increased the risk compared …
Tablets, Antidepressant, Effexor, Venlafaxine hydrochloride, Venlafaxine, Hydrochloride
Reference ID: 3101735 - Food and Drug …
www.accessdata.fda.govDocetaxel Injection in combination with doxorubicin and cyclophosphamide is indicated for the adjuvant treatment of patients with operable node-positive breast cancer.
Cytotec® - Food and Drug Administration
www.accessdata.fda.govcytotec® (misoprostol) warnings cytotec (misoprostol) administration to women who are pregnant can cause abortion, premature birth, or birth defects.
Administration, Drug, Food and drug administration, Food, Cytotec
Namenda XR (memantine hydrochloride) capsules …
www.accessdata.fda.govThe most commonly observed adverse reactions seen in patients administered NAMENDA XR in the controlled clinical trial, defined as those occurring at a frequency of at least 5% in the
Clinical, Capsule, Hydrochloride, Namenda xr, Namenda, Memantine hydrochloride, Memantine
FOR INTRAVENOUS USE ONLY - Food and Drug …
www.accessdata.fda.govNDA 50-706/S-022 Page 3 MERREM® I.V. (meropenem for injection) FOR INTRAVENOUS USE ONLY To reduce the development of drug-resistant bacteria and maintain the effectiveness of
Drug, Food, Food and drug, Only, Intravenous, For intravenous use only
NOT FOR USE IN NEONATES CONTAINS BENZYL …
www.accessdata.fda.govDepo-Medrol® methylprednisolone acetate injectable suspension, USP NOT FOR USE IN NEONATES CONTAINS BENZYL ALCOHOL . Not For Intravenous Use . DESCRIPTION
Alcohols, Neonates, Contains, In neonates contains benzyl, Benzyl, In neonates contains benzyl alcohol
HIGHLIGHTS OF PRESCRIBING INFORMATION …
www.accessdata.fda.govDacogen is indicated for treatment of patients with myelodysplastic syndromes (MDS) including previously treated and untreated, de novo and …
Venofer - Food and Drug Administration
www.accessdata.fda.govNDA 21-135/S-017 Page 3 Venofer ® (iron sucrose injection, USP) Rx Only DESCRIPTION Venofer® (iron sucrose injection, USP) is a brown, sterile, aqueous, complex of …
Administration, Drug, Food and drug administration, Food, Only, Venofer
Diphenhydramine Hydrochloride Injection
www.accessdata.fda.govHOW SUPPLIED Diphenhydramine Hydrochloride Injection, USP is a clear and colorless solution available as: 50 mg/mL in a 1 mL prefilled single-use syringe.
Related documents
Pediatric Urinary Tract Infections - Stanford Medicine
med.stanford.eduPediatric Urinary Tract Infections Steven L. Chang, MD, Linda D. Shortliffe, MDT Department of Urology, Stanford University School of Medicine, 300 Pasteur Drive, S-287, Stanford, CA 94305-2200, USA The urinary tract is a common site of infection in the pediatric population.
School, University, Medicine, Pediatric, Infections, Urinary, Tract, Urinary tract, Stanford, Stanford university school of medicine, Pediatric urinary tract infections
Guideline for Prevention of Catheter-Associated Urinary ...
www.cdc.govJun 06, 2019 · Guideline for Prevention of Catheter-Associated Urinary Tract Infections (2009) Last update: June 6, 2019 Page 8 of 61 I. Executive Summary This guideline updates and expands the original Centers for Disease Control and Prevention (CDC) Guideline for Prevention of Catheter -associat ed Urinary Tract Infections ( CA UTI ) publis hed in 1981.
Center, Control, Centers for disease control and prevention, Disease, Prevention, Infections, Urinary, Urinary tract infections, Tract
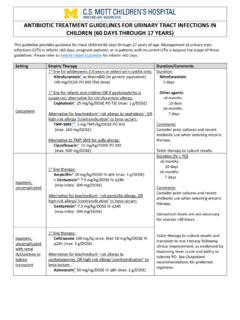
ANTIBIOTIC TREATMENT GUIDELINES FOR URINARY …
www.med.umich.eduANTIBIOTIC TREATMENT GUIDELINES FOR URINARY TRACT INFECTIONS IN CHILDREN (60 DAYS THROUGH 17 YEARS) This guideline provides guidance for most children 60 days through 17 years of age. Management of urinary tract infections (UTI) in infants <60 days, pregnant patients, or in patients with recurrent UTIs is beyond the scope of these guidelines.
Guidelines, Antibiotic, Treatment, Infections, Urinary, Urinary tract infections, Tract, Antibiotic treatment guidelines for urinary, Antibiotic treatment guidelines for urinary tract infections
Nosocomial Infections 2004 - Centers for Disease Control ...
www.cdc.govUTI, Urinary tract infection; BSI, bloodstream infection. * Number of urinary catheter-associated UTIs 31000 Number of urinary catheter-days y Number of central line-associated BSIs 31000 Number of central line-days z Number of ventilator-associated pneumonias 31000 Number of ventilator-days to construct tables comprised of data from fewer and
HIGHLIGHTS OF PRESCRIBING INFORMATION Before initiating ...
docs.boehringer-ingelheim.comWarnings and Precautions , Ketoacidosis (5.2) of urinary tract infections and tr 1/2020 -----INDICATIONS AND USAGE----- JARDIANCE is a sodium-glucose co-transporter 2 (SGLT2) inhibitor indicated: • as an adjunct to diet and exercise to improve glycemic control in adults
PedsCases Podcast Scripts
pedscases.cominclude urinary tract infections, meningitis and sepsis. Note that meningitis and pyelonephritis classically present with vomiting and fever. Neurological causes of vomiting relate to increased intracranial pressure and can include hydrocephalus, intracranial neoplasms, and pseudotumor cerebri in older children.
National Healthcare Safety Network (NHSN) Catheter ...
www.hospitalsafetygrade.orgassociated urinary tract infections (CAUTI) will be calculated among patients in the following patient care locations: • Intensive Care Units (ICUs) (excluding patients in neonatal ICUs [NICUs: Level II/III and Level III nurseries]) • Specialty Care Areas (SCAs) - …
Pediatric Nursing and Health Care - Carter Center
www.cartercenter.orgPediatric Nursing and child health care iv 3.2. Indications and use of naso-gastric tube 27 3.3. Care of child under specific conditions 32 3.4. Tracheostomy Care 36 Study Questions 40 CHAPTER FOUR: CARE OF THE NEW BORN 41 4.1. Care of normal new born 41 4.2. Causes of low birth weight 44 4.3. Perinatal and infant infections 51 4.4.
Health, Nursing, Care, Pediatric, Infections, Pediatric nursing and health care
Related search queries
Pediatric Urinary Tract Infections, Stanford University School of Medicine, Urinary tract, Pediatric, Urinary, Urinary tract infections, Centers for Disease Control and Prevention, ANTIBIOTIC TREATMENT GUIDELINES FOR URINARY, ANTIBIOTIC TREATMENT GUIDELINES FOR URINARY TRACT INFECTIONS, Infections, Pediatric Nursing and Health Care