www.isd622.org
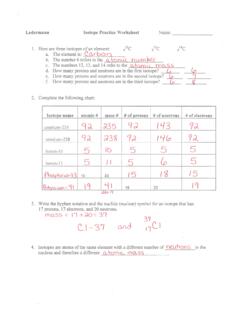
Ledermann: Name: On the Inside and Mass Part 1: Label the parts of this atom (nucleus, protons, electrons, neutrons) Part 2: Answer these: 1. 2.
Tags:
Information
Domain:
Source:
Link to this page:
Please notify us if you found a problem with this document:
Documents from same domain
www.isd622.org
www.isd622.orgDifferences Between Ionic and Covalent Compounds A compound is defined as the chemical combination of two or more elements. A chemical bond is the "glue" that holds atoms of different elements together. Bonds can be classified into two general types: ionic and covalent. Ionic bonds generally occur between a metallic atom and a nonmetallic atom.
Isotope Worksheet Answer Key - ISD 622
www.isd622.orgLedermann Name 1. Determine the average atomic mass of the following mixtures of isotopes. 128 127 126 a. t, 17%- 3% I (sðf' 8) 197 198 19 q. 5
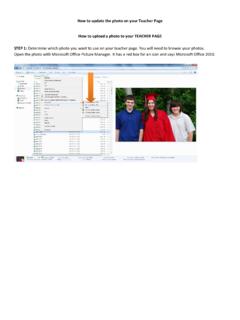
How to update the photo on your Teacher Page How to …
www.isd622.orgOpen the photo with Microsoft Office Picture Manager. It has a red box for an icon and says Microsoft Office 2010. How to update the photo on your Teacher Page The picture will open in a new window. Under FILE click on SAVE AS and give the photo a name so you know it is the one
Manager, Microsoft, Office, Pictures, Microsoft office, Microsoft office picture manager
19-21 Absolute Value Inequalities Real World …
www.isd622.org1921 Absolute Value Inequalities Real World Problems.notebook April 14, 2014 LT 3: I can represent realworld situations with equations, inequalities, and absolute value.
World, Value, Problem, Real, Equations, Inequalities, Absolute, Notebook, Absolute value, Absolute value inequalities real world, Absolute value inequalities real world problems
Approx. Reading Book Title Author Grade Level - ISD 622
www.isd622.orgDear Zoo Campbell, R. F 1.42 Dozen Dogs, A Ziefert, Harriet F 1.42 Foolish Goose Simon, Shirley F 1.42 Going for a Walk De Regniers, B. F 1.42 Harry Takes a Bath Ziefert, Harriet F 1.42 Have You Seen the Crocodile? West, Colin F 1.42 Here Comes A Bus Ziefert, Harriet F 1.42
Title, Reading, Book, Grade, Read, Authors, Dear zoo, Approx, Reading book title author grade
CHEMISTRY COMPUTING FORMULA MASS WORKSHEET
www.isd622.orgchemistry stoichiometry worksheet B. MASS - VOLUME and VOLUME - VOLUME PROBLEMS Since chemical equations for chemical reactions state the relative numbers of moles for each
Worksheet, Computing, Chemistry, Equations, Formula, Mass, Chemistry computing formula mass worksheet
10 Supreme Court Cases Every Teen Should Know
www.isd622.org10 Supreme Court Cases Every Teen Should Know The nation's highest court has had plenty to say about everything from free speech at school to teenagers' rights in the legal system. By Tom Jacobs For those of us on the outside, the U.S. Supreme Court can seem remote and mysterious. But the Court,
Approx. Reading Book Title Author Grade Level
www.isd622.orgBook Title Author Reading Level Approx. Grade Level Flying Crews, Donald E 1.28 Foot Book, The Seuss, Dr. E 1.28 Funny Man, A Jenson, Patricia E 1.28
Integer Operations Review Name: - ISD 622
www.isd622.orgInteger Operation Review Name: _____ _____ Adding Integers (+85) + (-96) = (-11) (+80) + (+57) = (+137) (+86) + (-38) = (+48) (+22) + (-41) = (-19) (-18) + (-45 ...
Mole to Mole Answer Key - ISD 622
www.isd622.orgStoichiometry: Multiple Choice [dentifr the choice that best completes the statement or answers the question. 1. Which branch of chemistry deals with the mass relationships of elements in compounds and the mass relationships
Related documents
Atoms and Their Parts - Amazon S3
s3.amazonaws.comWith the exception of hydrogen, all the atoms have three main parts. The parts of an atom are protons, electrons, and neutrons. A proton is positively charged and is located in the center or nucleus of the atom. All atoms of the same element have the same number of protons. The number of protons in the
s3.amazonaws.com
s3.amazonaws.comName Parts of the Atom ill in the ollowin table about the arts o the atom. (AMU stands or atomic mass unit.) LOCATION IN ATOM MASS (in AMU's) PART OF THE ATOM ELECTRIC CHARGE
Subatomic Particles Guided Notes Name: Period: Parts of ...
accelwarriorchem.weebly.com3. What is the mass number of this atom? 4. If an atom of carbon has a mass number of 13, how many neutrons does it have?
Notes, Name, Part, Periods, Guided, Particles, Mato, Subatomic, Subatomic particles guided notes name
Parts of an Atom •All matter is made of atoms. •Each and ...
ssichemistry.weebly.comParts of an Atom •All matter is made of atoms. •Each and every atom has three parts, called subatomic particles. Proton Neutron Electron. Parts of an Atom •Properties of Subatomic Particles Particle Symbol Location Relative Electric Charge Relative Mass Actual Mass (g)
Part, Particles, Three, Mato, Called, Subatomic, Parts of an atom, Three parts, Called subatomic particles
Chapter Outline Review of Atomic Structure
people.virginia.eduThe atomic mass of the 12C atom is 12 amu. The atomic weight of an element= weighted average of the atomic masses of the atoms naturally occurring isotopes. Atomic weight of carbon is 12.011 amu. The atomic weight is often specified in mass per mole.
Outline, Review, Chapter, Structure, Atomic, Mato, Chapter outline review of atomic structure
Atom Parts and Function - blogs.rsd13ct.org
blogs.rsd13ct.orgAtom Parts and Function. Parts of the Atom We have learned: Nucleus is the center of atom that contains: Protons - positive charge Neutrons - neutral, act like glue Overall charge of nucleus is positive Electron cloud/energy levels that contain: Electrons - negative charge.
Parts of an atom - Mr. Dodd's Science Class
doddscience.weebly.comName: Parts of the Atom Section: Part 1 - Label the parts of the atom below (protons, neutrons, electrons, nucleus, quarks), Part 2 - Answer the following questions,
List the 3 main types of ¾ subatomic particles and ...
www.austincc.edu3 13 Objective 2 Normally, the number of electrons in an atom equals the number of protons and the overall charge of the atom is zero. However, atoms may gain or lose electrons: ¾If an atom gains electrons, it will have an extra negative charge for each electron gained. ¾If an atom loses electrons, it will have an extra positive charge for each electron lost.
LESSON PLAN: Introducing the Atom - Periodic table
periodictable.rosendigital.comLESSON PLAN: Introducing the Atom periodictable.rosendigital.com Context Elements are substances that cannot be broken down into simpler forms of matter, and they are the primary constituents of all matter. The elements are the basis of all chemical interactions, and the …
Plan, Lesson plan, Lesson, Introducing, Mato, Introducing the atom
KM 654e-20150109102424
www.cpsk12.orgBohr Model Drawing Draw a Bohr model of a chlorine atom in the space below. Be sure to place the electrons in the correct orbitals and to fill out the key for the subatomic particles.