Functional Groups - Purdue University
I. Carboxylic Acids • contain a “carboxylic acid” group – a carbonyl (C=O) group bonded to a hydroxyl group at the carbonyl carbon atom • note that in condensed structural formulas, the carboxylic acid group may be written as –COOH
Download Functional Groups - Purdue University
Information
Domain:
Source:
Link to this page:
Please notify us if you found a problem with this document:
Documents from same domain
20.1 Mass Spectrometry Fundamentals - Purdue …
www.chem.purdue.edu20.1 Mass Spectrometry Fundamentals ... The mass spectrum can be used to determine molecular structure, to quantify single compounds or mixtures of
Fundamentals, Mass, Molecular, Spectrometry, 1 mass spectrometry fundamentals
Ch. 7 - Enolates - Purdue University
www.chem.purdue.edu651.06 209 Enolates Enolates are the conjugate anions of carbonyl compounds. Although they have been known and used since the turn of the 20th Century, it was the development of “specific
Enzyme Kinetics: Velocity - Purdue University
www.chem.purdue.edu• Enzyme activity can be assayed in many ways ... Enzyme Kinetics: Velocity . CHM333 LECTURES 15: 2/20/13 SPRING 2013 Professor Christine Hrycyna 105 • An example of how to do a ... • V is the reaction rate (velocity) at a substrate concentration [S]
Activity, Rates, Enzymes, Reactions, Kinetics, Velocity, Enzyme kinetics, Reaction rates
PURDUE UNIVERSITY INSTRUMENT VAN PROJECT
www.chem.purdue.eduFACTORS AFFECTING ENZYME ACTIVITY PURDUE UNIVERSITY INSTRUMENT VAN PROJECT QUESTIONS 1. In what way is the rate of the reaction dependent on the concentration of the enzyme?
Project, University, Activity, Rates, Instruments, Reactions, Purdue, Purdue university instrument van project, Activity purdue university instrument van project
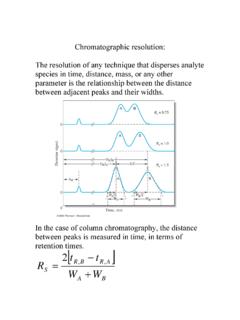
how to determine HETP from a peak - Purdue University
www.chem.purdue.eduChromatographic resolution is defined as the ratio of the difference of the retention times of two components to the sum of the peaks widths at the
The Fermentation of Pyruvate - Purdue University
www.chem.purdue.eduundergo lactate fermentation. Two pyruvates are converted to two lactic acid molecules, which ionize to form lactate. In this process two NADH + H+ are converted to two NAD+. Our muscle cells can undergo this process when they are in oxygen debt. If enough oxygen is not present to undergo aerobic respiration, pyruvate will undergo lactic acid
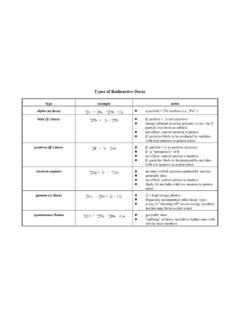
Types of Radioactive Decay - Purdue University
www.chem.purdue.eduTypes of Radioactive Decay type example notes alpha (α) decay 23 9 8 2 U 6 4 2 He + 23 9 4 0 Th + 2 0 0γ! α particle = 4 2 He nucleus (i.e., 4 2 He 2+) beta (β-) decay 23 9 4 0 Th 6! 0 1 e + 23 9 4 1 Pa! β-particle = ! 0 1 e (an electron)! energy released in decay process creates the β-particle (not from an orbital)! net effect: convert ...
Experiment 2: Acid / base titration - Purdue University
www.chem.purdue.eduExperiment 2: Acid / base titration cunknown =±62.0 0.5 mM @ 95% confidence level Nikolai Skrynnikov TA: Boone Prentice Section number: 1 25 Jan 2008 (data courtesy of Ike Fehrenbacher, 2004) 1
THEORY OF XRF - Purdue University
www.chem.purdue.eduXRF, and Chapter 4 describes how this physics is applied to spectrometers and their components. Chapter 5 explains how an XRF analysis is done. It describes the process of sample taking, measuring the sample and calculating the composition from the measurement results.
Purdue University: Department of Chemistry: Home
www.chem.purdue.eduSep 21, 2009 · Draw the structure of the peptide LCYRAIDCG as it would exist at pH 6.5 under oxidizing conditions. Be sure to draw any disulfide bonds that could form. EH — O-NA- Label the N-terminus of the peptide you drew in 3B above with a V. Label the C-terminus of the peptide you drew in 3B above with a Calculate the net charge of LCYRAIDG at pH 1.5.
Related documents
Synthesis, Isolation, and Purification of an Ester
www.dhouts.comacid, to form methyl acetate. The systematic name for acetic acid is ethanoic acid, and the systematic name for the ester product is methyl ethanoate. Experiment Overview In this experiment, a quantitative esterification reaction is performed. The process has three parts- reaction, isolation, and purification.
Acetic Acid
www.labchem.comSubstance name : Acetic Acid CAS-No. : 64-19-7 Product code : LC10100 Formula : C2H4O2 Synonyms : Acetic acid, glacial / alcohol of vinegar / carboxylic acid C2 / ethanoic acid / ethylic acid / methanecarboxylic acid / pyroligneous acid / vinegar acid . 1.2. Recommended use and restrictions on …
Ch 05 Carboxylic Acids and Esters - Angelo State University
www.angelo.edu• For molecules with two carboxylic acid groups the carbon chain in between the two carboxyl groups (including the carboxyl carbons) is used as the longest chain; the suffix -dioic acid is used. • For molecules with more than two carboxylic acid groups, the carboxyl groups are named as carboxylic acid substituents. C O HO C O OH ethanedioic ...
States, University, Acid, Carboxylic, Carboxylic acids, Angelo, Angelo state university
Synthesis of Carboxylic Acids
web.mnstate.edu• Carboxylic acids are completely converted to carboxylate salts by base • Carboxylate salts are completely neutralized back to carboxylic acids by strong acid • The resonanance stabilization makes carboxylates much more stable than hydroxide or alkoxide anions, which is why the parents are carboxylic “acids”
Carboxylic Acid Structure and Chemistry
webhome.auburn.eduThe carboxylic acid moiety is considered to be a highly polar organic functional group. This polarity results from the presence of a strongly polarized carbonyl (C=O) group and hydroxyl (O-H) group. Recall that oxygen is a relatively electronegative atom and when
Carboxylic Acids - Rutgers University
crab.rutgers.eduCarboxylic acid derivatives differ in the nature of the group bound to the acyl group. -OH is an acid-Cl is the acid chloride-OCOR' is the anhydride-OR' is the ester-NR 2 is the amide Nucleophilic acyl substitution can interconvert all of these different acid derivatives.
24. CARBOXYLIC ACID AND DERIVATIVES - MasterJEE Classes
masterjeeclasses.comFeb 08, 2019 · Chemistry | 24.5 (c) Melting Points: (i) Carboxylic acid with more than 8 carbon atom and a double bond have low melting point due to the inability to form a stable lattice. High m.p. HC 3 COOH Lie on opposite side Low m.p. CH 3 COOH Lie on same side (ii) Structures with even number of C atoms have a higher melting point as compared to structures having odd …
Acid, Derivatives, Carboxylic, Carboxylic acids, Carboxylic acid and derivatives