PURDUE UNIVERSITY INSTRUMENT VAN PROJECT
FACTORS AFFECTING ENZYME ACTIVITY PURDUE UNIVERSITY INSTRUMENT VAN PROJECT QUESTIONS 1. In what way is the rate of the reaction dependent on the concentration of the enzyme?
Tags:
Project, University, Activity, Rates, Instruments, Reactions, Purdue, Purdue university instrument van project, Activity purdue university instrument van project
Information
Domain:
Source:
Link to this page:
Please notify us if you found a problem with this document:
Documents from same domain
Enzyme Kinetics: Velocity - Purdue University
www.chem.purdue.edu• Enzyme activity can be assayed in many ways ... Enzyme Kinetics: Velocity . CHM333 LECTURES 15: 2/20/13 SPRING 2013 Professor Christine Hrycyna 105 • An example of how to do a ... • V is the reaction rate (velocity) at a substrate concentration [S]
Activity, Rates, Enzymes, Reactions, Kinetics, Velocity, Enzyme kinetics, Reaction rates
20.1 Mass Spectrometry Fundamentals - Purdue …
www.chem.purdue.edu20.1 Mass Spectrometry Fundamentals ... The mass spectrum can be used to determine molecular structure, to quantify single compounds or mixtures of
Fundamentals, Mass, Molecular, Spectrometry, 1 mass spectrometry fundamentals
Ch. 7 - Enolates - Purdue University
www.chem.purdue.edu651.06 209 Enolates Enolates are the conjugate anions of carbonyl compounds. Although they have been known and used since the turn of the 20th Century, it was the development of “specific
how to determine HETP from a peak - Purdue University
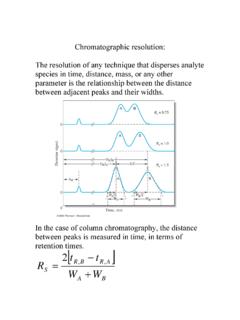
www.chem.purdue.eduChromatographic resolution is defined as the ratio of the difference of the retention times of two components to the sum of the peaks widths at the
The Fermentation of Pyruvate - Purdue University
www.chem.purdue.eduundergo lactate fermentation. Two pyruvates are converted to two lactic acid molecules, which ionize to form lactate. In this process two NADH + H+ are converted to two NAD+. Our muscle cells can undergo this process when they are in oxygen debt. If enough oxygen is not present to undergo aerobic respiration, pyruvate will undergo lactic acid
Types of Radioactive Decay - Purdue University
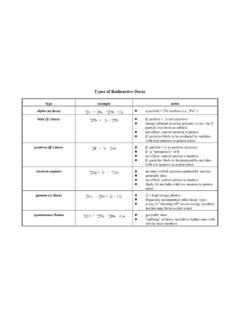
www.chem.purdue.eduTypes of Radioactive Decay type example notes alpha (α) decay 23 9 8 2 U 6 4 2 He + 23 9 4 0 Th + 2 0 0γ! α particle = 4 2 He nucleus (i.e., 4 2 He 2+) beta (β-) decay 23 9 4 0 Th 6! 0 1 e + 23 9 4 1 Pa! β-particle = ! 0 1 e (an electron)! energy released in decay process creates the β-particle (not from an orbital)! net effect: convert ...
Experiment 2: Acid / base titration - Purdue University
www.chem.purdue.eduExperiment 2: Acid / base titration cunknown =±62.0 0.5 mM @ 95% confidence level Nikolai Skrynnikov TA: Boone Prentice Section number: 1 25 Jan 2008 (data courtesy of Ike Fehrenbacher, 2004) 1
THEORY OF XRF - Purdue University
www.chem.purdue.eduXRF, and Chapter 4 describes how this physics is applied to spectrometers and their components. Chapter 5 explains how an XRF analysis is done. It describes the process of sample taking, measuring the sample and calculating the composition from the measurement results.
Functional Groups - Purdue University
www.chem.purdue.eduI. Carboxylic Acids • contain a “carboxylic acid” group – a carbonyl (C=O) group bonded to a hydroxyl group at the carbonyl carbon atom • note that in condensed structural formulas, the carboxylic acid group may be written as –COOH
Purdue University: Department of Chemistry: Home
www.chem.purdue.eduSep 21, 2009 · Draw the structure of the peptide LCYRAIDCG as it would exist at pH 6.5 under oxidizing conditions. Be sure to draw any disulfide bonds that could form. EH — O-NA- Label the N-terminus of the peptide you drew in 3B above with a V. Label the C-terminus of the peptide you drew in 3B above with a Calculate the net charge of LCYRAIDG at pH 1.5.
Related documents
Biology Ivestiation The effect of pH on Enzyme Activity
s3.amazonaws.comBiology Ivestiation The effect of pH on Enzyme Activity Sangam bharti Kyiv International School ... What is the effect of different pH concentration on the rate of enzyme activity? Hypothesis: The rate of enzyme activity of catalase will be the greatest at the optimum pH of 7. ... concentration of the pH is increased the rate of reaction starts ...
Activity, Rates, Reactions, Effect, Biology, Rate of reaction, Biology ivestiation the effect of, Ivestiation
Lab Activity H13 Food Coloring Kinetics
webs.anokaramsey.eduLab Activity H13 Food Coloring Kinetics OUTCOMES After completing this lab activity, the student should be able to: explain how the concentration of a reactant affects the reaction rate. determine the effect of temperature of a reaction mixture on the reaction rate.
Activity, Food, Rates, Reactions, Kinetics, Coloring, Reaction rates, Lab activity h13 food coloring kinetics
Kinetics Unit Activity - Chemical Kinetics KEY
sparks.cm.utexas.eduKinetics Unit Activity - Chemical Kinetics KEY The purpose of this activity is to develop concepts of chemical kinetics: Specifically to look at reaction rate laws. First, it is important to understand rates. Consider the following reaction C H 3 C l + OH-o C H 3 OH + C l ...
Chemical, Activity, Unit, Rates, Reactions, Kinetics, Reaction rates, Kinetics unit activity chemical kinetics key
Effect of pH on Catalase Activity - The Pingry School
faculty.pingry.k12.nj.usEffect of pH on Catalase Activity Tommie S. Hata The Pingry School Biology Mr. Hata Investigation date: September 23, 2002 Submitted: September 24, 2002 ... The reaction rate of the enzyme will then be measured and graphed. It is expected that the reaction rate …
Activity, Rates, Reactions, Effect, Catalase, Reaction rates, Effect of ph on catalase activity
Amylase: a sample enzyme - Bio 111 and 112 Home Page
intro.bio.umb.eduRATE OF REACTION (100/TIME TO ENDPOINT) 10% 5% 1% Note: We are calculating the rate of the reaction (3rd column above) by assuming that we started ... group will pick one of these and investigate its effect on the activity of amylase.
SAM Teachers Guide Chemical Reactions and Stoichiometry
ri-itest.concord.orgSAM Teachers Guide Chemical Reactions and Stoichiometry ... • Analyze how concentration and temperature affect reaction rate. ... This activity is supported by both ...
Guide, Chemical, Activity, Rates, Teacher, Reactions, Stoichiometry, Reaction rates, Sam teachers guide chemical reactions and stoichiometry
The Effects of Exercise on Reaction Time - Spring Issue
jass.neuro.wisc.eduresting heart rate, blood pressure, and reaction time were 70, 130, and .224 seconds, respectfully. The post-exercise heart rate, blood pressure, and reaction time were 169, 128/70, and 0.169 seconds, respectfully for the female subject and 145, 140, 0.216 for the male subject.
Exercise, Time, Rates, Reactions, Effect, Effects of exercise on reaction time
Kruthi Renduchintala J0519 - California State Science Fair
cssf.usc.eduKruthi Renduchintala The Effect of Enzyme Concentration on the Reaction Rate J0519 Objectives/Goals Enzymes are organic catalysts that speed up reactions by decreasing the activation energy needed to start the chemical reaction. Therefore, my hypothesis is that as the enzyme concentration increases, the speed
Rates, Reactions, Reaction rates, Kruthi renduchintala, Kruthi, Renduchintala
Catalase Kinetics Chris Su Meiyi Li TR
web.mit.edu! 3! measuring the changes in pressure inside the reaction vessel over time. From approximating ideal conditions and using the PV=nRT equation, one can obtain the rate of the reaction being 1/RT d[P]/dt.3 Another important equation in enzyme kinetics is the Michaelis-Menten mechanism.
Rates, Reactions, Kinetics, Rich, Catalase, Catalase kinetics chris su meiyi, Meiyi
Enzyme Kinetics: Properties of â-Galactosidase
biology.kenyon.edudetermine the reaction rate, and design and conduct and experiment on the potential interaction between pH and temperature on the sensitivity of this enzyme. You will be using â-galactosidase
Related search queries
Biology Ivestiation The effect of, Activity, Rate, RATE OF REACTION, Lab Activity H13 Food Coloring Kinetics, Reaction rate, Reaction, Kinetics Unit Activity - Chemical Kinetics KEY, Effect of pH on Catalase Activity, Amylase, SAM Teachers Guide Chemical Reactions and Stoichiometry, Effects of Exercise on Reaction Time, Kruthi Renduchintala, Catalase Kinetics Chris Su Meiyi