Acid base titration
Found 9 free book(s)7 Acid-Base Titration Computer
www2.vernier.comComputer 7 Advanced Chemistry with Vernier 7 - 1 Acid-Base Titration A titration is a process used to determine the volume of a solution that is needed to react with a
AB titration expt - Oneonta
employees.oneonta.eduACID-BASE TITRATION Form A The Molar Mass of an Unknown, Diprotic Acid Titration is the process for ascertaining the exact volume of one solution that is chemically equivalent to a given amount of another substance,
DETERMINING THE CONCENTRATION OF CITRIC …
www.flavours.asia3 data and analysis sheet: determining the concentration of citric acid in a soft drink using acid/base titration name: _____
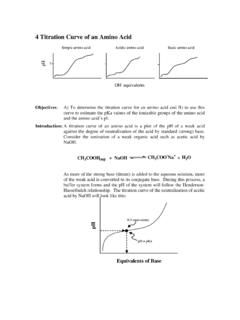
4 Titration Curve of an Amino Acid - Xavier …
www.xula.eduWhen a weak monoprotic acid is titrated by a base, a buffer system is formed. The pH of this system follows the Henderson-Hasselbalch equation:
ACID-BASE TITRATIONS - cffet.net
cffet.netChapter 3 17 ACID-BASE TITRATIONS The reaction of acid and base involves transfer of a proton from acid to base and is often called a neutralisation reaction.
Titration of Oxalic Acid Lab - Sardis Secondary School
www.sardissecondary.caPart B: Titration: 10. Place the flask with acid and phenolphthalein under the buret. The buret tip should be down about 1cm inside the mouth of the flask to avoid losing any of the base.
25 Titration Diprotic Acid - Vernier Software & …
www2.vernier.comTitration of a Diprotic Acid: Identifying an Unknown Chemistry with Vernier 25 - 3 5. Obtain approximately 60 mL of ~0.1 M NaOH solution in a 250 mL beaker.
Acid-base Equilibria and Calculations
www.chem1.comAcid-base Equilibria and Calculations A Chem 1 Reference Text Stephen K. Lower Simon Fraser University Contents 1 Proton donor-acceptor equilibria 4 1.1 The ion product of water ..... 4
TITRATION OF A COLA PRODUCT - Fantastic Flavours
www.flavours.asia1 Experiment TITRATION OF A COLA PRODUCT The CCLI Initiative Computers in Chemistry Laboratory Instruction LEARNING OBJECTIVES The objective of this laboratory experim ent is to determine the molar co ncentratio n of phosphoric ac id in a