Aromatic Amines
Found 10 free book(s)Ch 06 Amines and Amides - Angelo State University
www.angelo.edu• Aromatic amines: named as derivatives of the parent compound aniline. Substituents attached to the nitrogen are indicated by using “N-” as the location number. NH2 aniline. Chapter 6 Amines and Amides 7 Examples: Nomenclature of Amines
NOMENCLATURE IN ORGANIC CHEMISTRY
scilearn.sydney.edu.auJ. Amines and Ammonium Salts 22 5. AROMATIC COMPOUNDS 23 (i) General Notes 23 (ii) Aromatic Hydrocarbons 23 (iii) Substituted Aromatic Hydrocarbons 24 2 A. Halogen and Nitro- Substituted Aromatics 24 B. Carboxylic Acids and Derivatives 24 C. Phenols and Thiophenols 25 D. Aldehydes and Ketones 26 ...
13 UnitUnitUnit - NCERT
ncert.nic.inAromatic primary amines cannot be prepared by this method because aryl halides do not undergo nucleophilic substitution with the anion formed by phthalimide. 6. Hoffmann bromamide degradation reaction Hoffmann developed a method for preparation of primary amines by
unit 13 - NCERT
www.ncert.nic.inAromatic primary amines cannot be prepared by this method because aryl halides do not undergo nucleophilic substitution with the anion formed by phthalimide. 6.Hoffmann bromamide degradation reaction Hoffmann developed a method for preparation of primary amines by
Incompatibilities of concentrated nitric acid: Never mix ...
ccc.chem.pitt.edu7 Amines, Aliphatic and Aromatic H H GT 7 E Explosion 8 Azo Compounds, Diazo Compounds and Hydrazines H G H GT H H H 8 P Violent Polymerization 9 Carbamates H G H GT G H 9 S Solubilization of toxic substance 10 Caustics H H G 10 U May be hazardous, but Unknown 11 Cyanides GT ...
Gas-chromatography/mass spectrometry (GC -MS ...
www.ccic.ohio-state.eduAug 17, 2015 · Aromatic Hydrocarbons Halocarbons Mercaptans Sulfides CS2 . Long lifetime and very low bleed at high operating temperatures. Temperature range: -60 °C to 350 °C . Ethers . Ketones . Aldehydes . Esters . Tertiary amines . Nitro compounds without α-H atoms . Nitrile compounds without α-H atoms . Column specifically tested for low -bleed ...
Naming Rules for Organic Compounds
web.viu.caAmines (RNH 2, R 2NH, R 3N) -many common names are still in usage -for simple primary amines, name as follows eg. CH 3CH 2CH 2CH 2NH 2 2-butanamine -for simple 2 o and 3 amines, name the substituents as N-alkyl groups eg. CH 3CH 2NHCH 3 N-methylethanamine -for molecules where the -NH 2 group is treated as a substituent, it is referred to as an ...
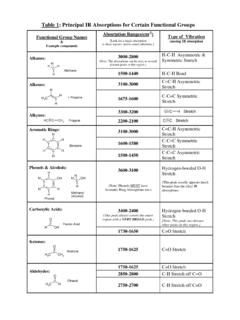
Table 1: Principal IR Absorptions for Certain Functional ...
academics.wellesley.eduAmines—Primary: N H H Ethylamine 1640-1560 N-H Bend 3500-3100 (ONE PEAK!) Amines—Secondary: N-H Stretch N CH3 H N-Methylethylamine 1550-1450 N-H Bend Nitriles: C C N H H H Methanenitrile 2300-2200 CNStretch 1600-1500 N=O Stretch Nitro Groups: H C 3 N + O O Nitromethane (Note: Both peaks are <200 cm-1 apart.) 1400-1300 N=O Bend
Structure Determination of Organic Compounds
bionmr.unl.eduErno Pretsch¨ · Philippe Buhlmann¨ · Martin Badertscher Structure Determination of Organic Compounds Tables of Spectral Data Fourth, Revised and Enlarged Edition 123
Rapid Revision Course for JEE Main 2022 Lecture Plan
dmf76jm51vpov.cloudfront.netS.No. Subject Chapter Name No. of Lectures 1 Physics Kinematics 3 2 Physics NLM & Friction 3 3 Physics Circular Motion & WPE 3 4 Physics COM 3