I stat cartridge
Found 9 free book(s)CLIA Waived Tests and CPT Codes - Home :: Washington …
www.doh.wa.govAbbott i-STAT 6+ Cartridge (WB) i-STAT Corporation 84520QW Abbott i-STAT CHEM8+ Cartridge (WB) i-STAT Corporation 84520QW Arkray SPOTCHEM EZ Chem Analyzer (WB) Arkray, Inc. 84520QW Calcium Abaxis Piccolo Blood Chemistry Analyzer WB (Chem 13 Panel, CMP, BMP, Renal) Abaxis, Inc. 82310QW
PROCEDURE MANUAL FOR THE i-STAT SYSTEM
clinlab.uams.eduThe i-STAT System incorporates comprehensive components needed to perform blood analysis at the point of care. The system consists of the following primary components: i-STAT 1 Analyzer When a sample-filled i-STAT cartridge is inserted into the i …
PROCEDURE MANUAL FOR THE i-STAT SYSTEM - Lagaay
www.lagaay.comi-STAT cartridge is inserted into a handheld for analysis, the handheld automatically controls all functions of the testing cycle including fluid movement within the cartridge, calibration and continuous quality monitoring. A PCx Plus Glucose Test Strip is scanned and inserted into the i-STAT 1 Analyzer (Model Number 300) and a drop of
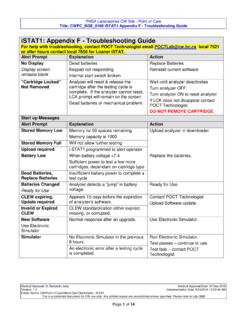
iSTAT1: Appendix F - Troubleshooting Guide
policyandorders.cw.bc.caProcedure: i-STAT Ceramic Conditioning Cartridge (CCC) for Analyzer Pin Conditioning REFERENCES. PHSA Laboratories CW Site - Point of Care Title: CWPC_BGE_0160 iSTAT1 Appendix F - Troubleshooting Guide Medical Approval: Dr Benjamin Jung Medical Approval Date: 22 …
PCO2 AND CALCULATED VALUES FOR HCO3, TCO2, BASE …
harvardapparatus.comshould be retested using another cartridge. Intended Use The test for PCO2, as part of the i-STAT System, is intended for use in the in vitro quantification of carbon dioxide partial pressure in ar te ri al, venous, or capillary whole blood. Contents Each i-STAT cartridge contains one reference electrode (when potentiometric sensors are included
PO2 AND CALCULATED OXYGEN SATURATED
www.harvardapparatus.comanother cartridge. Intended Use The test for PO2, as part of the i-STAT System, is intended for use in the in vitro quantification of oxygen partial pressure in ar te ri al, venous, or capillary whole blood. Contents Each i-STAT cartridge contains one reference electrode (when potentiometric sensors are included in
Frequently Asked Questions FAQs Abbott i-STAT
www.cms.govApr 29, 2020 · 2/28/2020 and 4/9/2020, respectively. With respect to the G3+ (BLUE) test cartridge, CMS is exercising enforcement discretion (as of 3/27/2020) to allow laboratories with a Certificate of Registration that applied for a Certificate of Compliance, or laboratories with a Certificate of Compliance, that have the i-STAT system, to use the
HEMATOCRIT/HCT AND CALCULATED HEMOGLOBIN/HB
www.abaxis.comanother cartridge. Intended Use The test for hematocrit, as part of the i-STAT® System, is intended for use in the in vitro quantification of packed red blood cell volume fraction in arterial, venous, or capillary whole blood. Hematocrit measurements can aid in the determination and monitoring of normal or abnormal total red
Automotive Data Solutions Inc. INSTALL GUIDE COM-BLADE …
images.idatalink.comNov 19, 2020 · CARTRIDGE INSTALLATION 1 Slide cartridge into unit. Notice button under LED. 2 Ready for Module Programming Procedure. MODULE PROGRAMMING PROCEDURE 1 Insert key into ignition. 2 Turn key to START position. START 5 Module Programming Procedure completed. 4 OFF Turn key to OFF position. 3 Wait, LED will turn solid BLUE for 2 seconds.