Search results with tag "Genotoxic"
Risk Assessment and Management of Genotoxic Impurities …
www.nihs.go.jpGenotoxic Impurities The focus of this guideline is on DNA reactive substances which can be detected by Ames assay. Application of Threshold of Toxicological Concern (TTC) to control genotoxic impurities. Risk assessment for patients and healthy volunteers during clinical development. Evaluation of genotoxicity of impurities using the
Q&A on the CHMP Guideline on the Limits of Genotoxic ...
www.ema.europa.eupotentially genotoxic impurities or to higher levels of previously recognized potentially genotoxic impurities then the situation should be discussed with the competent authority. Question 9. What is a reasonable policy for setting specifications for potentially genotoxic
ICH guideline M7 on assessment and control of DNA reactive ...
www.ema.europa.euNo. The terms “mutagenic potential” and “genotoxic potential” are not interchangeable. Mutagenic potential refers to the ability of a compound to induce point mutations (i.e., bacterial reverse mutation assay), while genotoxic potential refers to both mutagenic and clastogenic potential. ICH M7 focuses specifically on
Risk Assessment and Management of Genotoxic Impurities in ...
www.nihs.go.jpRisk Assessment and Management of Genotoxic Impurities in Pharmaceuticals Masamitsu Honma, Ph.D. Division of Genetics and Mutagenesis, National Institute of Health Sciences,
N-Nitrosodimethylamine (NDMA) - World Health …
www.who.intThere is also ample evidence that NDMA is genotoxic both in vivo and in vitro. Activation by liver microsomal S9 fractions is necessary for a positive in vitro result. The recent observation that human S9 fractions are much more active in promoting genotoxicity in the Ames test than rat S9 fractions suggests that humans may be especially ...
A C DNA R (M P L P C R M7(R1) - ICH
database.ich.orgsome cases where a drug substance intended for other indications is itself genotoxic at therapeutic concentrations and may be expected to be associated with an increased cancer risk. Exposure to a mutagenic impurity in these cases would not significantly add to the
Q3C (R5) Impurities: guideline for residual solvents
www.tga.gov.auqualification of impurities as expressed in the guideline for drug substance (Q3A, Impurities in New Drug Substances) or drug product (Q3B, ... Non-genotoxic animal carcinogens or possible causative agents of other irreversible toxicity such as neurotoxicity or teratogenicity.
London, 28 June 2006 EMEA/CHMP/QWP/251344/2006 …
www.ema.europa.euThe toxicological assessment of genotoxic impurities and the determination of acceptable limits for such impurities in active substances is a difficult issue and not addressed in sufficient detail in the existing ICH Q3X guidances. The data set usually available for …
NONCLINICAL EVALUATION FOR ANTICANCER …
database.ich.orgapplications for pharmaceuticals that are genotoxic and target rapidly dividing cells (e.g., crypt cells, bone marrow) in general toxicity studies or belong to a class that has been well characterized as causing developmental toxicity.
Essential IND Strategies: Fundamental Considerations on ...
health.ucdavis.edudrug formulations, with well characterized impurity profiles. 5) ... Does not address potential genotoxic impurities in API Genetic Toxicology . Requirements ICH Core Battery: ... Small molecule – commonly stand alone studies Biological – …
Chemistry, Manufacturing, and Controls of Drug Candidates ...
accelerate.ucsf.eduGenotox Impurities* •“exposure to the potentially genotoxic impurities can not exceed 60 micrograms per day. For longer duration clinical trials the levels would have to be further reduced; for clinical trials of greater than one year duration, the daily exposure to these impurities should not exceed 1.5 micrograms.”
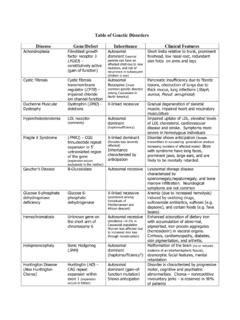
Table of Genetic Disorders - Loyola University Chicago
www.meddean.luc.eduenvironmental genotoxic agents. Title: Microsoft Word - Table of Genetic Disorders.doc Author: afrankf Created Date: 4/21/2005 9:59:39 PM ...
This document was valid from 1 September 2008 to 1 June ...
www.ema.europa.euidentified as acceptable for genotoxic impurities in pharmaceuticals by the CHMP. 2. DEFINITION AND SCOPE . Metal catalysts and metal reagents are defined here as chemical substances that are used to change the rate of chemical reactions or which act on other chemical substances in chemical reactions. For the
Guideline on assessment and control of DNA reactive ...
www.ema.europa.euof toxicological concern (TTC) for genotoxic carcinogens, resulting in an acceptable intake of 0.025 µg/kg bw/day. For target animals, the approaches to derive acceptable intakes are specified in section 8. Guideline on assessment and control of DNA reactive (mutagenic) impurities in veterinary medicinal products
Introduction to ICH - The Quality Guidelines – An Overview
admin.ich.orgQ 3 – Impurities Q 4 – Pharmacopoeias Q 5 – Biotechnological Products ... residual solvents in pharmaceuticals for the safety of patients, recommends use of less ... Non-genotoxic animal carcinogens or possible causative agents of other irreversible toxicity.
Genotoxic Impurities and Its Risk Assessment in Drug …
lupinepublishers.comGenotoxic impurities can get incorporated into drug substances through the various sources, the major source is the starting material used in the synthesis of drug substances and its impurities. Similarly, genotoxic intermediate and by-products formed in the
Genotoxic Impurities in Pharmaceuticals - InTech - Open
cdn.intechopen.com17 Genotoxic Impurities in Pharmaceuticals Abolghasem Jouyban 1 and Hamed Parsa 2 1Drug Applied Research Center and Faculty of Pharmacy, 2Tuberculosis and Lung Disease Research Center, Tabriz University of Medical Sciences, Tabriz, Iran 1. Introduction Genotoxic compounds induce genetic mutation s and/or chromosomal rearrangements and
Genotoxic impurities in small molecule drug candidates
www.toxicology.orgAssessment of Genotoxic Impurities in Small Molecule Drug Candidates John Wisler, PhD, DABT Kurt A Black, Ph.D, DABT Comparative Biology and Safety Sciences, Amgen Inc.
Similar queries
Assessment and Management of Genotoxic, Genotoxic, Assessment, Genotoxic impurities, Of Genotoxic Impurities in Pharmaceuticals, World Health, Impurities, Of genotoxic impurities, Pharmaceuticals, Drug, Small molecule, Table of Genetic Disorders, Loyola University Chicago, Genotoxic Impurities in Pharmaceuticals, Reactive (mutagenic) impurities in veterinary medicinal products, Introduction to ICH - The Quality Guidelines – An Overview, In pharmaceuticals, Genotoxic Impurities in Small Molecule Drug