Search results with tag "Visible"
ULTRAVIOLET AND VISIBLE SPECTROSCOPY
www.uobabylon.edu.iqUltraviolet and Visible Spectroscopy This absorption spectroscopy uses electromagnetic radiations between 190 nm to 800 nm and is divided into the ultraviolet (UV, 190-400 nm) and visible (VIS, 400-800 nm) regions. Since the absorption of ultraviolet or visible radiation by a molecule leads transition among electronic
Standard Operating Procedure Ultraviolet Visible (UV-Vis ...
engineering.purdue.edu1 Standard Operating Procedure Ultraviolet–Visible (UV-Vis) Spectroscopy in POWER Laboratory Lizbeth Rostro, May 2012 Description of Process The ultraviolet–visible spectroscopy (UV-Vis) utilizes light to determine the absorbance or transmission of a chemical species in either solid or aqueous state. Personal Protective Equipment
Making Thinking Visible - Harvard University
www.pz.harvard.eduSuch routines jump-start thinking and make it visible. Thinking routines are one element of an initiative called Visible Thinking that we, our colleagues at Project Zero, and collaborators in various schools have developed. In our research, we have explored the practicality of using thinking routines and documentation as
BULLETIN BOARDS THAT MAKE LEARNING VISIBLE
www.makinglearningvisibleresources.orgwork. They can also make learning visible. Why: Bulletin boards reflect a class or school’s identity. In and outside the classroom, bulletin boards that make learning visible show what we value, offer opportunities for reflection, help learners make connections within and across subject matter, and contribute to
Flame Test Lab Activity Key - University of South Florida
natasha.eng.usf.edudifferent kinds of light. This light may be visible or invisible, infrared or ultraviolet radiation. Analysis of this light can be used to identify elements and molecules. In today’s lab, we will observe the visible range of light emission. Objectives 1. Perform a flame test to identify the characteristic color of metal ions. 2.
UV -Visible spectroscopy - IIT Delhi
web.iitd.ac.inUV -Visible spectroscopy Absorption of light in the UV/Visible part of the spectrum (210 ± 900 nm). The transitions that result in the absorption of electromagnetic radiation in this region of the spectrum are transitions between electronic energy levels. Generally, the most probable transition is …
THINKING ROUTINES FOR MAKING THINKING VISIBLE
gslps.vic.edu.auTHINKING ROUTINES FOR MAKING THINKING VISIBLE Routines for Introducing and Exploring Ideas Routines for Synthesising and Organising Ideas ... • Makes learning visible by focusing to big ideas and if students can put • Questions generate a richer level …
Module 2 Spectroscopic techniques Lecture 3 Basics of ...
www.nptel.ac.iniv. UV/Visible region: UV and visible regions are involved in the electronic transitions in the molecules. The spectroscopic methods using UV or visible light therefore come under ‘Electronic spectroscopy’. v. X-ray radiation: X-rays are high energy electromagnetic radiation and causes transitions in the internal electrons of the molecules.
ALARM VALVE - NAFFCO FZCO
www.naffco.comThe alarm valve must be installed in a readily visible and accessible location and provision to be made in such a way that alarm line drain is visible and accessible. 3. Where water pressure fluctuates, the variable pressure ... placing a roving fire patrol in the area covered by the system. The patrol should continue until the system is back
The Four Capabilities of Leadership - Saylor Academy
resources.saylor.orgCommunicate constantly, says Professor Ancona. By doing so, a leader is visible to his or her employees and can be a reassuring presence. Being visible is a key trait of good managers and executives, it shows workers and customers that the organization’s leadership is committed to, and actively overseeing its current projects.
Reducing Contamination Risks of Compressed Air in Food ...
www.foodengineeringmag.comair is invisible. It leaves no visible trace where it contacts the food, other food contact surfaces, or the packaging. Without adequate hurdles and physical barriers in place the microbial, particulate, and (in some cases) compressor oil contamination is left behind after the air dissipates. Some examples of direct and indirect contact points are:
2.9.19. PARTICULATE CONTAMINATION: SUB-VISIBLE …
uspbpep.com2.9.20. Particulate contamination: visible particles EUROPEAN PHARMACOPOEIA 6.0 Wet the inside of the filter holder fitted with the membrane filter with several millilitres ofparticle-free water R.Transfer to the filtration funnel the total volume of a solution pool or of a single unit, and apply vacuum. If needed, add stepwise
Evidence on zero-tolerance policing The New York crime drop
www.justiceinspectorates.gov.ukvisible (see The effectiveness of visible police patrol). References Bottoms, A. (2012) Developing Socio-Spatial Criminology, in Maguire, M., Morgan, R. and Reiner, R. (eds) The Oxford Handbook of Criminology (fifth edition). Oxford: Oxford University Press. Bowling, B. (1999) The Rise and Fall of New York Murder: Zero Tolerance or Crack‟s ...
MEASURING AND LEVELING TOOLS
www.farnell.comRead-out: visible and audible signals Power: 9V battery IntelliSensor Technical specification: Detection depth: up to 19 mm Detects: wood, metal in dry walls Detection accuracy: +/- 3 mm Read-out: visible and audible signals Power: 9V battery Easy to read LCD display One button operation - easy to use Deep Read feature Detects hot electrical ...
Standards of Human Comfort - Texas Architecture
soa.utexas.edux-rays ultra infrared radar shortwave violet 100 150 200 300 500 750 1000 1500 2000 electromagnetic spectrum 1022 1020 1018 1016 1014 1012 1010 108 106 104 frequency in hertz (cycle per second) Firgure 06. Visible Light and the Elecomagenetic Spectrum
ANALYSIS OF PLANT PIGMENTS USING PAPER …
www.chem.purdue.educolor: UV violet blue green yellow red IR <-----> 300 400 500 600 700 800 high energy light low energy light Spectrophotometers and related devices can "sense" not only visible light but other wavelengths such as ultraviolet as well.
Professional Development to Practice Hattie’s Ten Mind ...
www.moedu-sail.orgThis feedback allows teachers to see learning through the eyes of their students. It makes learning visible and facilitates the planning of next steps. The feedback that students receive from their teachers is also vital. It enables students to progress towards challenging learning intentions and goals (Waak, 2013) Teacher-Student Relationships
MARINE CORPS TATTOO POLICY
www.marines.milvisible in the properly fitting PT uniform. (2) Concept of Operations. All tattoos must be in compliance with this Bulletin as described below. ... Ultra-Violet (UV) Tattoos. Any tattoo only
Thin Layer Chromatography (TLC) - Boston College
www.bc.edufew minutes, then circle any visible spots with a pencil. Into a filter paper lined 400 mL beaker, pour either pure isopropanol or 1:1 isopropanol: concentrated ammonia to a depth of about 0.5 cm. Develop the second plate in the same manner as the first using the chosen solvent. Mark the solvent front and circle the spots.
The 5E Instructional Model: A Learning Cycle Approach for ...
files.eric.ed.govvisible (National Research Council [NRC], 2000). Recently, Project 2061: Science ... learning cycles, the one that will be highlighted here as a method to support inquiry-based teaching is the 5E Instructional Model (Bybee & Landes, 1990). The 5E Instructional Model
Thermal Infrared Remote Sensing - IIIT
researchweb.iiit.ac.in• The main difference between thermal infrared and near infrared is that thermal infrared is emitted energy, whereas the near infrared is reflected energy, similar to visible light. Thermal Remote Sensing Atmospheric Transmission The windows normally used for aircraft platforms are in the 3-5 micron and 8-14 micron wavelength regions
2.2.46. CHROMATOGRAPHIC SEPARATION TECHNIQUES …
www.drugfuture.comUltraviolet/visible (UV/Vis) spectrophotometers and flame ionisation detectors are the most commonly employed detectors. Light scattering detectors, infrared absorption spectrophotometers, thermal conductivity detectors or other special detectors may be used. METHOD Prepare the test solution(s) and the reference solution(s) as prescribed.
A Summary Report - Police Foundation
www.policefoundation.orgevaluation the value of visible police patrol. The year-long experiment tested the effectiveness of the traditional police strategy of routine preventive patrol and sought to determine whether the resources in the Kansas City, Missouri, Police Department ordinarily allocated to preventive patrol could safely be devoted to
ColorOS 7.1 User Guide - OPPO
ipics.oppo.comname and password for your hotspot. When "Visible to Other Devices" is turned off, you can manually add networks to connect to the hotspot Up to 10 devices can connect to your hotspot at the same time. Wi-Fi Tethering cannot be used simultaneously with OPPO Share, Wi-Fi Direct, Multi-screen Interaction, and other functions. USB Tethering
TIE-35: Transmittance of optical glass - University of Arizona
wp.optics.arizona.eduTIE-35: Transmittance of optical glass DATE October 2005 PAGE 3/12 Figure 2-1: Measured optical constants of fused silica (SiO 2 glass). The influence of the UV-edge on the refractive index is clearly visible. [1] Regions of strong dispersion, with steep slopes in the refractive index curve, correspond to
CONSTAT AMIABLE D’ACCIDENT AUTOMOBILE ACCIDENT …
www.lolivier.frYES NO In the affirmative : patrol or Police Station of Material damage other than to vehicles A and B (nature and extend, name and address of owner) Estimate of damage Name and address of the garage where the vehicle can be visible OUI NON YES NO OUI NON YES NO Wounded name Age Address Occupation
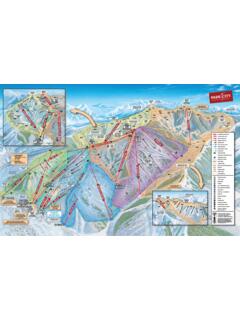
ParkCity Trail Map - Ski
images.ski.commountain patrol ticket office restrooms lockers snowboard school kids signature jupiter east face scotts bowl o-zone p-zone molly's mcconkevs bowl georgeÂnn terrain parks kings crown 3 at mght) little kings ... terrain is visible from the resort base area. resort
Solar Radiation Hand Book (2008) - India Environment Portal
www.indiaenvironmentportal.org.into 3000nm. About half of the radiant energy is in the visible short-wave part of the electromagnetic spectrum. The other half is mostly in the near-infrared part, with some in the ultraviolet part of the spectrum. Solar radiation having wavelength less than 0.286nm (called ultraviolet) is absorbed by ozone layer in stratosphere.
LE GUIDE COMPLET DU DÉCRET TERTIAIRE
www.unis-immo.frpublier cette attestation à un endroit visible et accessible, permettant un accès aisé à l’information. Cette attestation comporte un visuel permettant de savoir où en est chaque bâtiment dans sa démarche d'amélioration. 1 Informations sur son patrimoine 2 Consommations annuelles d'énergie finale 3 Consommation d’énergie finale de
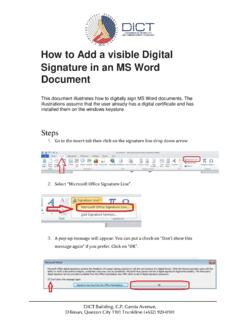
How to Add a visible Digital Signature in an MS Word …
dict.gov.phMicrosoft Office digital signatures combine the familiarity of a paper signing experience with the convenience of a digital format. While this feature provides users with the ability to verify a documents integrity, evidentary laws may vary by jurisdiction. Microsoft thus cannot warrant a digital signature'slegal enforceability. The third-party
LTFRB | LTFRB Official Website
ltfrb.gov.phgiven suitable and audible signal. 15. Increasing speed vThen being overtaken- increasing the speed of the motor vehicle before the overtaking vehicle has completely passed. 16. Overtaking when left side is not visible or clear of traffic- driving to the left side of the center line of a highway in overtaking or passing another vehicle
P.O. Box 2120
www.doe.virginia.govTeachers are encouraged to go beyond the standards as ... Mathematical communication becomes visible where learning involves participation in mathematical discussions. ... instruction that addresses individual learning needs, maximizing the opportunity to learn.
Effective teacher behaviours
webcontent.ssatuk.co.ukthe key to making teaching effective was through ‘visible ... scaffold thinking. Develops social skills. Pupils’ articulating ideas develops higher order skills. Many pupils ... It is also important to have routines and systems for starts and ends of lessons, transitions,
Short-wave Infared Imagery (SWIR) - European Space Imaging
www.euspaceimaging.comare not possible with visible and near-infrared alone • Bands optimized for the detection of features of greatest interest • Ideal for penetrating smoke, mapping minerals and identifying man-made features SWIR Specifications Spectral Characteristics SWIR 1 1195 - 1225 nm SWIR 2 1550 - 1590 nm SWIR 3 1640 - 1680 nm SWIR 4 1710 - 1750 nm
Running head: POLICE AGENCIES AND RELAXED GROOMING ...
www.cji.educontrol room officer, dispatcher, patrol officer, student patrol coordinator, investigator, and patrol sergeant. Currently, ndhe is assigned the role of 2 ... grooming policy that restrict facial hair to above the lip and no visible tattoos since the early ... Little Rock police are now fully staffed; However, keeping it that way is difficult ...
Lecture 7: Propagation, Dispersion and Scattering
topex.ucsd.edu¥ For visible light, water vapor, dust, and other particles ranging from a few tenths of a micrometer to several micrometers in diameter are the main scattering agents. The amount of scatter is greater than Rayleigh scatter and the wavelengths scattered are longer. ¥ Pollution also contributes to beautiful sunsets and sunrises. The
Lecture 6: Thermal Radiation
topex.ucsd.edu3.Thermal infrared: emitted, can only be detected using electro-optical sensors (3.0 - 5.0 and 8 - 14 µm). Microwave Radar sensors, wavelengths range from 1mm - 1m (Ka, Ku, X, C, S, L & P) Spectral bands Three important spectral bands in remote sensing: ¥visible light ¥infrared radiation ¥microwave radiation Image from NASA 1987.
Teaching writing: Guide for Years 1–3
www.qcaa.qld.edu.auwrite using both a pencil and the voice. Drawings, symbols, letters and numerals are produced to communicate a message. Talk surrounds their written graphics. Some young writers may begin by selecting an idea, an experience or a thought to be put into print and discovering some strategy, such as drawings, for making that thought visible. Others
CITY OF NEW YORK PARKS & RECREATION SEASONAL JOB …
www.nycgovparks.org• Under supervision, perform patrols of park facilities as part of a highly visible uniformed division. Ensure the safety and enjoyment of park users and the protection and stewardship of parks property. • Issue summonses for the violation of park rules and regulations; may detain or arrest violators of City and State laws.
INSTRUMENTAL ANALYSIS (I)
www.uotechnology.edu.iqVisible enough energy for valence (bonding) excitations . UV and x-ray enough energy for core (inner) excitations ... Inorganic ions can be quantitatively determined by spectrophotometry by reaction with some ligands to produce colored complexes which have clear absorption in …
Visible Learning for Teachers - hozir.org
hozir.orgVisible Learning for Teachers John Hattie’s ground-breaking book Visible Learningsynthesized the results of more than 15 years research involving millions of students and represented the biggest ever collection
Visible Emissions Field Manual, EPA Methods 9 and 22
www3.epa.govsions from unconfined sources other than opacity meth- ods. Method 22 Since EPA promulgated Method 22 in 1982, it has be- come an important tool in the conaol of visible emis- sions. Method 22 is a qualitative technique that checks only the presence or …
Visible Learning: The Five Strands - Midlothian
www.midlothian.gov.ukVisible Learning school, it is a process of self - evaluation of the five strands. Schools can determine which areas to develop and which key outcomes to evaluate by gathering evidence and working collaboratively to identify where they are and planning where they want to be. ...
Similar queries
Visible, Ultraviolet Visible, Ultraviolet–visible, MAKING THINKING VISIBLE, Routines, Thinking, THINKING ROUTINES, Visible Thinking, Using thinking routines, Ultraviolet, MAKING THINKING VISIBLE Routines, Spectroscopy, Alarm valve, Patrol, Leadership, Contamination, PARTICULATE CONTAMINATION, 2.9.20. Particulate contamination: visible particles, The effectiveness of visible police patrol, Audible, Ultra, Violet, Spectrophotometers, Professional Development to Practice Hattie, Teachers, Learning, Learning visible, Boston College, Infrared, A Summary Report, Police, Of visible police patrol, The effectiveness, Your, To your, Transmittance, Glass, ParkCity, Solar Radiation Hand Book 2008, Add a visible Digital Signature in an, Digital, Digital signature, Maximizing, Making, Spectral, Lecture 6: Thermal Radiation, Using, Spectrophotometry, Visible Learning, Visible Emissions, Sources